(UroToday.com) The 2022 GU ASCO Annual meeting included a session on novel treatment implementation focusing on PSMA targeting and beyond, with a presentation by Dr. Michael Hofman discussing PSMA PET and FDG PET as predictors of response and prognosis in the TheraP trial.1 This trial previously showed that 177Lu-PSMA-617 improved PSA≥50% response rate (PSA50-RR), PSA-PFS, and radiographic PFS (rPFS) compared with cabazitaxel in mCRPC progressing after docetaxel. TheraP study inclusion required high PSMA uptake (SUVmax ≥ 20) and no lesions that were FDG positive and PSMA negative. At the 2022 GU ASCO meeting, Dr. Hofman reported on PSMA PET and FDG PET as potential predictive and prognostic biomarkers. The hypotheses were that (i) increased PSMA intensity would lead to an increased response to 177Lu-PSMA-617 vs cabazitaxel, and (ii) increased FDG volume would lead to decreased response to both 177Lu-PSMA-617 and cabazitaxel.
This study prospectively analyzed semi-automated quantitative PET parameters in centrally-collected 68Ga-PSMA-11 PET and 18F-FDG PET in 200 eligible men. SUVmean ≥ 10 on PSMA PET was evaluated as a predictive biomarker for response to 177Lu-PSMA-617 vs cabazitaxel. Metabolic tumor volume ≥ 200mL on FDG PET was tested as a prognostic biomarker accounting for the randomly assigned treatment. Quantitative PET cut-offs were pre-specified from prior research.2 Responses were defined according to PSA50-RR (primary endpoint), PSA-PFS, and rPFS. Binary and PFS endpoints were analyzed using logistic and Cox regression, respectively.
Very high PSMA uptake on PSMA PET (SUVmean ≥ 10) was seen in 35/99 (35%) assigned 177Lu-PSMA-617 and 30/101 (30%) assigned cabazitaxel. The odds of a response to 177Lu-PSMA-617 vs. cabazitaxel were significantly higher for men with SUVmean ≥ 10 (OR 12.2, 95% CI 3.4-59 vs. 2.2, 95% CI 1.1-4.5; p = 0.03). In men with SUVmean ≥ 10, the PSA50-RR for 177Lu-PSMA-617 vs. cabazitaxel was 32/35 (91%) vs. 14/30 (47%). In men with PSMA SUVmean < 10, the PSA50-RR were 33/64 (52%) vs. 23/71 (32%):
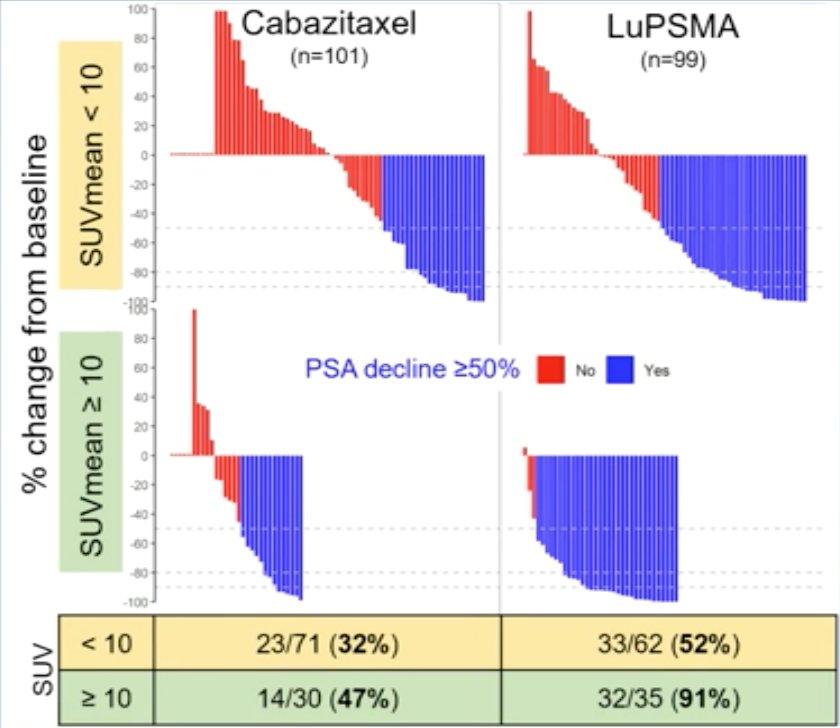
The HR for PSA-PFS for 177Lu-PSMA-617 vs cabazitaxel was 0.45 (95% CI 0.25-0.80) for SUVmean ≥ 10 vs. 0.77 (95% CI 0.53-1.12) for SUVmean < 10 (p = 0.2):

High-volume metabolic disease on FDG PET (metabolic tumor volume ≥200mL) was seen in 30/99 (30%) assigned 177Lu-PSMA-617 and 30/101 (30%) assigned cabazitaxel. The PSA50-RR in these men was 17/30 (57%) for 177Lu-PSMA-617 vs. 6/30 (20%) for cabazitaxel. In comparison, the PSA50-RR for men with metabolic tumor volume < 200mL on FDG PET was 48/69 (70%) for 177Lu-PSMA-617 vs. 31/71 (44%) for cabazitaxel. After accounting for treatment, the odds of a PSA50-response was lower among men with high metabolic tumor volume (OR 0.44; p = 0.01):

The HRs for high metabolic tumor volume on FDG PET adjusted for treatment were 1.44 (95% CI 1.03-2.02) for PSA-PFS (p = 0.03) and 1.79 (95% CI 1.28-2.52) for rPFS (p < 0.001):

There are several clinical implications for this study including (i) 177Lu-PSMA-617 should be prioritized in men with very high PSMA expression, (ii) quantitative PET/CT parameters are valuable, and (iii) more work is needed to automate workflows.
Dr. Hofman concluded his biomarker presentation based on data from the TheraP trial with the following take-home messages:
- In men with mCRPC, PSMA SUVmean ≥ 10 was predictive of a higher likelihood of favorable response to 177Lu-PSMA-617 than cabazitaxel
- A high volume of disease on FDG PET (metabolic tumor volume > 200 mL) was associated with a worse prognosis regardless of randomly assigned treatment.
Clinical trial information: NCT03392428.
Presented by: Michael S. Hofman, MBBS, FRACP, Peter MacCallum Cancer Center, Melbourne, Australia
Co-Authors: James P. Buteau, Andrew James Martin, Louise Emmett, Amir Iravani, Shahneen Kaur Sandhu, Anthony M. Joshua, Alison Yan Zhang, Roslyn J. Francis, Andrew Mark Scott, Arun Azad, Margaret Mary McJannett, Martin R. Stockler, Scott Williams, Ian D. Davis
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022
References:
- Hofman MS, Emmett L, Sandhu S, et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): A randomized, open-label, phase 2 trial. Lancet. 2021 Feb 27;397(10276):797-804.
- Ferdinandus J, Violet J, Sandhu S, et al. Prognostic biomarkers in men with metastatic castration-resistant prosate cancer receiving [177Lu]-PSMA-617. Eur U Nucl Med Mol Imaging. 2020 Sep;47(10):2322-2327.

