(UroToday.com) The 2022 GU ASCO Annual meeting included a urothelial carcinoma session featuring work from Dr. Peter O'Donnell and colleagues presenting results assessing the impact of primary tumor location on efficacy and safety of pembrolizumab in patients with locally advanced or metastatic urothelial carcinoma enrolled in the phase 2 KEYNOTE-052 and phase 3 KEYNOTE-045 trials. Pembrolizumab showed antitumor activity in first-line and second line for patients with urothelial carcinoma in the single-arm, phase 2 KEYNOTE-052 study1 and the randomized phase 3 KEYNOTE0452 study, respectively. This post hoc exploratory analysis evaluated whether primary tumor location affected efficacy and safety of pembrolizumab (KEYNOTE052; KEYNOTE-045) and chemotherapy (KEYNOTE-045).
KEYNOTE-052 enrolled cisplatin-ineligible patients with advanced/metastatic urothelial carcinoma who had not previously received systemic therapy; they received pembrolizumab (200 mg IV Q3W). KEYNOTE045 enrolled patients with advanced/metastatic urothelial carcinoma who had received platinum-containing chemotherapy; patients were randomly assigned 1:1 to receive pembrolizumab (200 mg IV Q3W) or investigator’s choice of chemotherapy (paclitaxel, docetaxel, or vinflunine). Both studies required patients to have measurable disease per RECIST v1.1. The study schema for these two trials is as follows:

Upper tract urothelial carcinoma included primary tumors in the renal pelvis or ureter; lower tract urothelial carcinoma included primary tumors in the bladder or urethra. Patients with upper tract and lower tract disease (upper tract/lower tract) were classified as lower tract. Patients receiving pembrolizumab were treated until disease progression, unacceptable toxicity, or withdrawal of consent, for up to 2 years. End points were PFS, ORR, and DOR per RECIST v1.1 by central radiology assessment and OS.
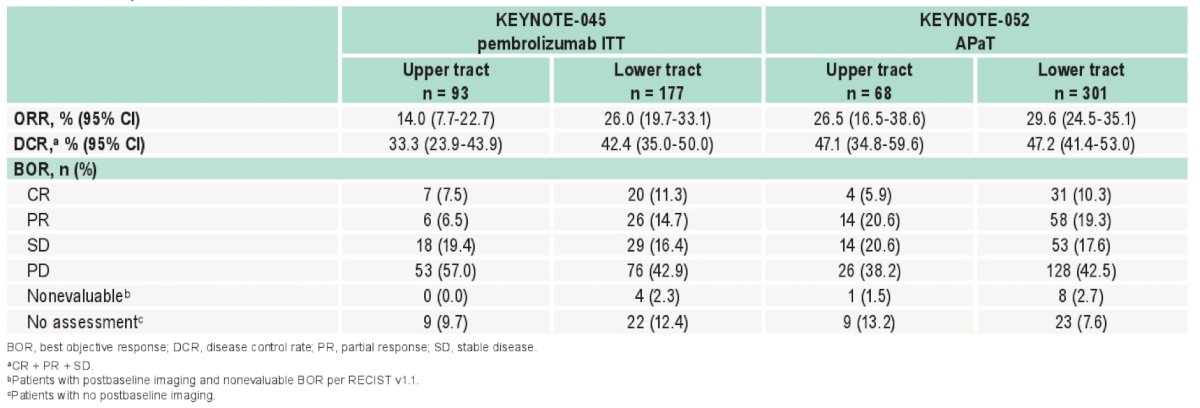
There were a total of 369 pembrolizumab-treated patients (68 upper tract; 301 lower tract [79 upper tract/ lower tract]) from KEYNOTE-052, plus 270 pembrolizumab-treated patients (93 upper tract; 177 lower tract [33 upper tract/lower tract]) and 272 chemotherapy-treated patients (94 upper tract; 178 lower tract) from KEYNOTE-045 evaluated. Median follow-up from randomization to data cutoff (September 26, 2020 and October 1, 2020, respectively) was ≥56 mo. Both studies enrolled a similar percentage of patients with PD-L1–positive tumors (25%-30%). PFS, ORR, DOR, and OS for pembrolizumab were consistent regardless of tumor location, although ORR for KEYNOTE-045 was lower for the upper tract group:
Overall survival by primary tumor location in the two trials was comparable and is highlighted in the following Kaplan-Meier curves:
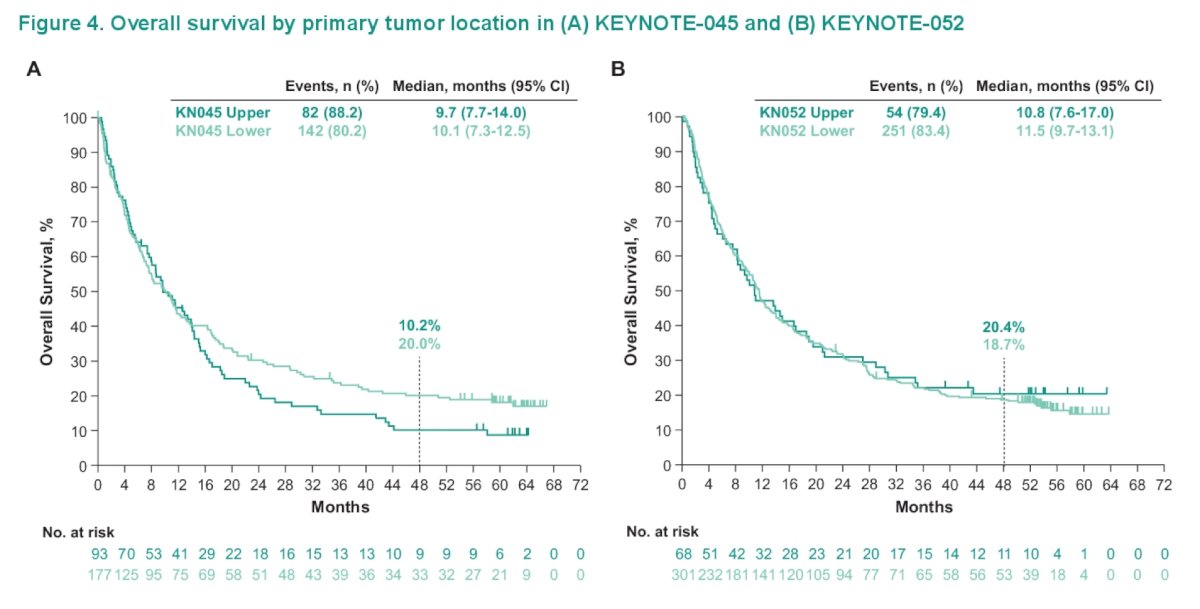
In the chemotherapy arm of KEYNOTE-045, similar efficacy was observed regardless of tumor location or regimen. Grade 3-5 treated related adverse events occurred at similar rates in KEYNOTE-052 (19.1% upper tract; 21.6% lower tract) and KEYNOTE-045 (17.2% upper tract; 16.8% lower tract).
Dr. O’Donnell concluded his presentation of the pooled analysis results of KEYNOTE-052 and KEYNOTE-045 assessing the impact of tumor location with the following take-home messages:
- OS, PFS, ORR, and DOR were generally consistent between KEYNOTE-045 and KEYNOTE-052 regardless of primary tumor location, although ORR was numerically lower for patients with upper tract disease than for those with lower tract disease in KEYNOTE-045
- Pembrolizumab showed manageable safety in patients regardless of primary tumor location
- These data suggest that patients with urothelial carcinoma may benefit from pembrolizumab regardless of primary tumor location
Co-Authors: Arjun Balar, Daniel E. Castellano, Ronald De Wit, David J. Vaughn, Thomas Powles, Jacqueline Vuky, Jae-Lyun Lee, Yves Fradet, Joaquim Bellmunt, Lawrence Fong, Daniel P. Petrylak, Winald R. Gerritsen, David I. Quinn, Stephane Culine, Dean F. Bajorin, Jin Zhi Xu, Kentaro Imai, Blanca Homet Moreno, Petros Grivas
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022
References:
- Balar AV, Castellano D, O’Donnell PH, et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): A multicentre, single-arm, phase 2 study. Lancet Oncol 2017;18(11):1483-1492.
- Bellmunt J, de Wit R, Vaughn DJ, et al. Pembrolizumab as Second-Line Therapy for Advanced Urothelial Carcinoma. N Engl J Med 2017;376(11):1015-1026.

