(UroToday.com) The 2022 GU ASCO Annual meeting included a urothelial carcinoma trials in progress session featuring the Double Antibody Drug Conjugate (DAD) phase I trial, presented by Dr. Bradley McGregor, assessing sacituzumab govitecan plus enfortumab vedotin for metastatic urothelial carcinoma progressing on platinum-based chemotherapy and PD1/L1 inhibitors. There have been multiple advances in therapy of advanced urothelial carcinoma following platinum and immune checkpoint blockade. Enfortumab vedotin is an antibody drug conjugate against Nectin-4 for urothelial carcinoma following platinum and immune checkpoint inhibitor or one line of therapy with ORR up to 52%. Sacituzumab govitecan is an antibody drug conjugate to SN38 against Trop-2 in urothelial carcinoma following progression in platinum and immune checkpoint inhibitor with ORR of 27%.1 Sacituzumab govitecan and enfortumab vedotin are both FDA approved in this space, though durations of response are short and overall survival limited at < 13 months. Given different mechanisms of action, non-overlapping toxicities and potential for synergy, enfortumab vedotin and sacituzumab govitecan is hypothesized to have manageable toxicities and demonstrate additive anti-tumor activity.
NCT04724018 is a single-center (Dana-Farber/Harvard Cancer Center), open-label, non-randomized phase I trial using a Bayesian Optimal Interval design. Sacituzumab govitecan and enfortumab vedotin agents are administered intravenously (IV) on days 1 and 8 every 3 weeks (1 cycle) until progression or intolerable toxicities. Laboratory evaluation is performed on days 1 and 8 every cycle with additional blood collected throughout treatment for pharmacokinetic and antidrug antibody studies. Imaging is performed at baseline and every 6 weeks for the first 4 cycles followed by every 9 weeks thereafter. Up to 4 dose levels of the combination of sacituzumab govitecan and enfortumab vedotin are studied with a starting dose of sacituzumab govitecan of 8 mg/kg and enfortumab vedotin of 1 mg/kg (dose level 1) with additional dose escalations outlined as follows:
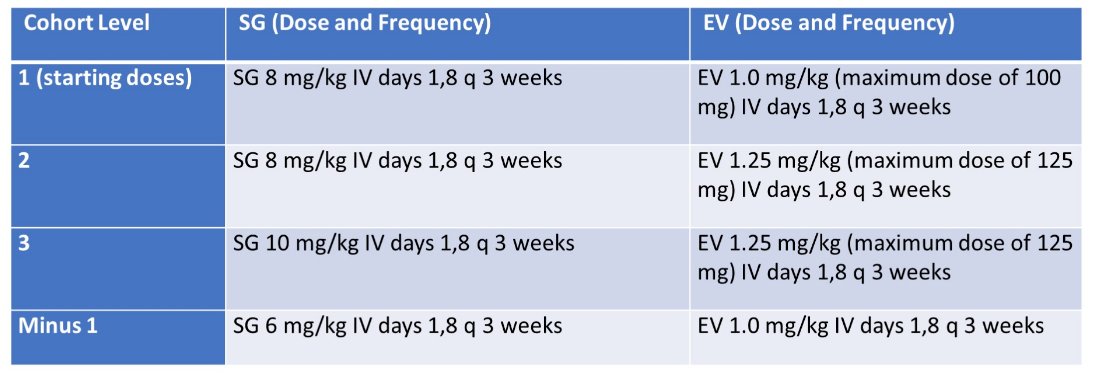
Dose de-escalation of sacituzumab govitecan to 6 mg/kg with the starting dose of enfortumab vedotin 1 mg/kg (dose level -1) is permitted. A total of 3 to 18 patients are expected to be enrolled in current dose level depending on dose limiting toxicities (DLTs). DLT is assessed during cycle 1 of therapy and toxicities are captured up to 4 weeks after the last dose of therapy and being defined by ≥1 of the following: neutropenic fever, thrombocytopenic bleeding, grade 3 neuropathy, of any duration and any other grade ≥3 non-hematologic toxicity to include hyperglycemia (except alopecia) lasting >1 week or requiring >3 weeks interruption of therapy or dose reduction. The study schema for this trial is as follows:
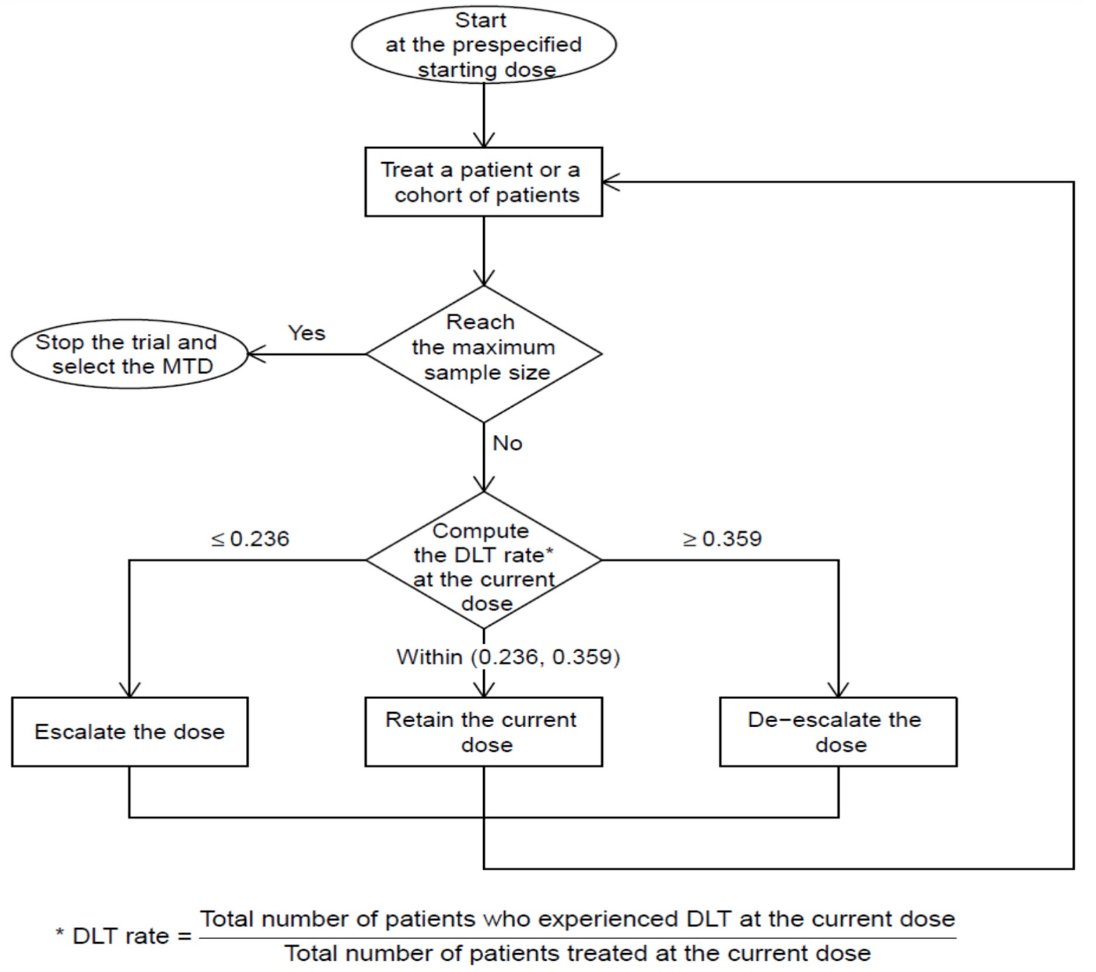
The first patient per 3-patient cohort is required to complete 1 full cycle (3 weeks) before additional patients can be enrolled in that cohort. Therefore, up to 24 patients may be accrued. Trial continues to enroll at dose level one.
Dr. McGregor emphasized that the DAD trial is a single arm phase 1 study evaluating enfortumab vedotin with sacituzumab govitecan in patients with metastatic urothelial carcinoma progression on immunotherapy and platinum therapy, or one therapy and ineligible for cisplatin.
Presented By: Bradley A. McGregor, MD, Dana-Farber Cancer Institute, Boston, MA
Co-Authors: Lucia Kwak, Charlene Mantia, Arvind Ravi, Jacob E Berchuck, Praful Ravi, Wenxin Xu, Martha Tuff, Joaquim Bellmunt, Xin Gao, Rajitha Sunkara, Guru P. Sonpavde
Affiliations: Dana Farber Cancer Institute, Boston, MA, Lank Center for Genitourinary Oncology, Dana-Farber Cancer Institute, Boston, MA, Beth Israel Deaconess Medical Center, Boston, MA, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, Massachusetts General Hospital, Boston, MA, Beverly Hosp, Lynnfield, MA
Written By: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022
References:
- Tagawa ST, Balar AV, Petrylak DP, et al. Metastatic urothelial carcinoma progressing after platinum-based chemotherapy and checkpoint inhibitors. J Clin Oncol. 2021 Aug 1;39(22):2474-2485.

