(UroToday.com) The 2022 GU ASCO Annual meeting included a urothelial carcinoma trials in progress session featuring the TROPHY-U-01 cohort 4 trial, presented by Dr. Scott Tagawa, assessing sacituzumab govitecan in combination with cisplatin in platinum-naïve patients with metastatic urothelial cancer. Cisplatin-based combination chemotherapy regimens with gemcitabine or methotrexate, vinblastine, and doxorubicin are commonly used as initial treatment for metastatic urothelial carcinoma. About 40-50% of patients achieve an objective response, but with limited duration of response and modest survival with these regimens:
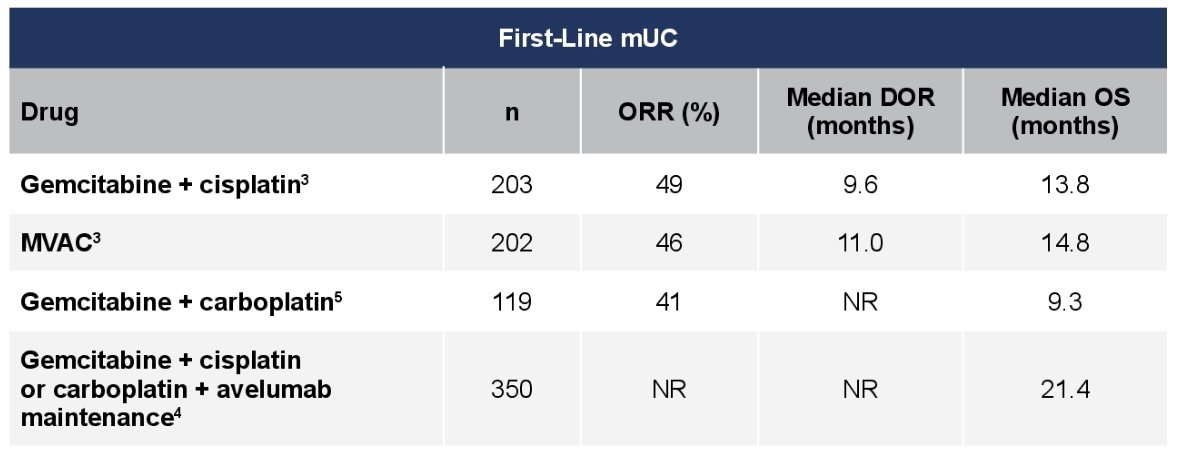
Alternative cisplatin-based combinations with improved efficacy while maintaining tolerability are needed. Sacituzumab govitecan is an antibody-drug conjugate composed of an anti-trophoblast cell-surface antigen-2 (Trop-2) antibody coupled to SN-38 (a topoisomerase-I inhibitor) via a proprietary hydrolysable linker. The mechanism of action concentrations active drug within the tumor and in the tumor microenvironment:
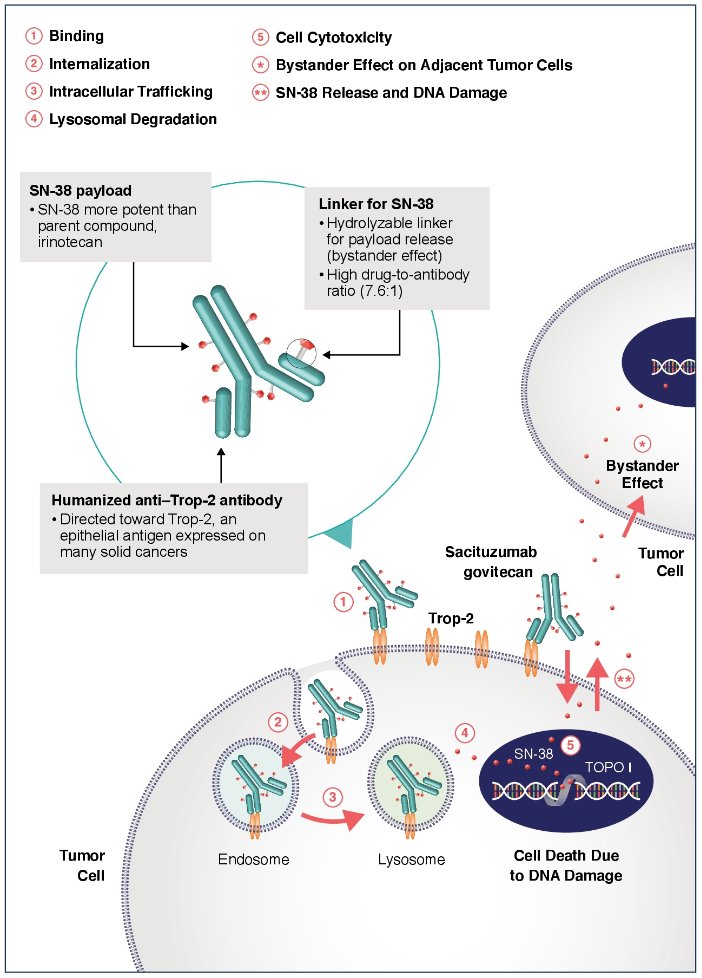
In vitro/in vivo urinary bladder cancer models suggest that the combination of sacituzumab govitecan and cisplatin resulted in additive antitumor activity over either agent alone (data on file). In the phase 2 registration TROPHY-U-01 study, sacituzumab govitecan monotherapy resulted in a 27% objective response rate (ORR) and a median overall survival (OS) of 10.9 months with a manageable, mostly non– cisplatin-overlapping toxicity profile in heavily pretreated patients with metastatic urothelial carcinoma [1]. These results led to accelerated approval of sacituzumab govitecan by the FDA in 2021 for patients with locally advanced or metastatic urothelial carcinoma who previously received platinum-containing chemotherapy and a checkpoint inhibitor. The hypothesis for this trial is that sacituzumab govitecan and cisplatin in combination may improve efficacy/safety over available first-line metastatic urothelial carcinoma regimens. TROPHY-U-01 Cohort 4 study will evaluate the safety, tolerability, and clinical activity of this novel combination.
TROPHY-U-01 (NCT03547973) is a multicohort, open-label, global phase 2 trial. Cohort 4 will evaluate combination sacituzumab govitecan and cisplatin in platinum-naive patients with metastatic urothelial carcinoma or unresectable locally advanced disease. Key eligibility requirements include Eastern Cooperative Oncology Group performance status 0–1, no prior anticancer monoclonal antibody or antibody-drug conjugate therapy within 4 weeks of study drug initiation, no history of active interstitial lung disease or non-infectious pneumonitis, and adequate hematologic, hepatic, and renal function. Patients will be treated with cisplatin at 70 mg/m2 on day 1 of a 21-day cycle (if creatinine clearance [CrCl] ≥60 mL/min) or at a split dose of cisplatin at 35 mg/m2 on days 1 and 8 of a 21-day cycle (if CrCl 50-59 mL/min) followed by sacituzumab govitecan (5, 7.5, or 10 mg/kg) on days 1 and 8 of a 21-day cycle. The recommended phase 2 dose will be the dose in which ≤30% dose-limiting toxicities are noted in cycle 1. Combination therapy will continue for up to 6 cycles, followed by sacituzumab govitecan and avelumab maintenance given until progression. Prophylactic granulocyte colony-stimulating factor is not allowed in cycle 1 of the safety lead-in phase. The study design for TROPHY-U-01 cohort 4 is as follows:

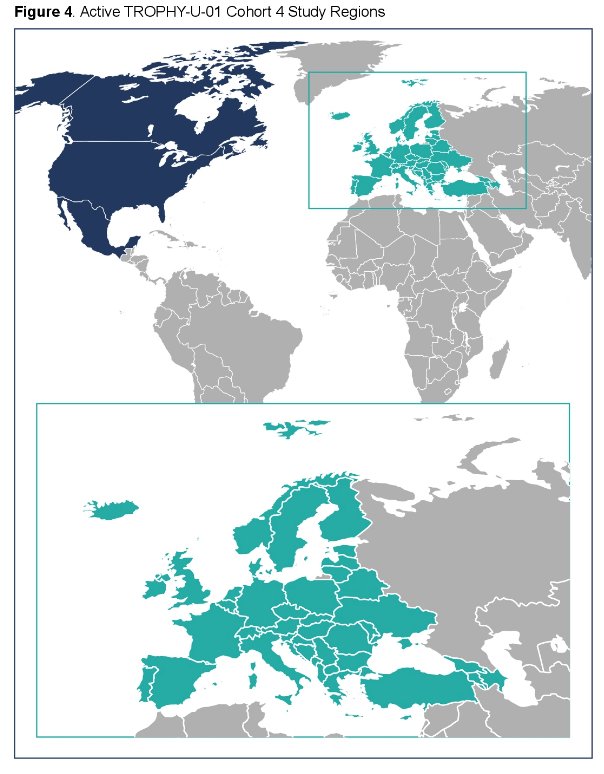
Primary endpoint is ORR per RECIST v1.1 by blinded independent central review (BICR). Secondary endpoints include progression-free survival, duration of response, and clinical benefit rate per BICR and investigator assessment. OS and safety will be assessed. Enrollment began in May 2020 and is ongoing, expanding to ~60 sites in North America and Europe:
Co-Authors: Petros Grivas, Daniel P. Petrylak, Cora N. Sternberg, Umang Swami, Astha Bhatia, Cabilia Pichardo, Trishna Goswami, Yohann Loriot
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022
References:
- Tagawa ST, Balar AV, Petrylak DP, et al. Metastatic urothelial carcinoma progressing after platinum-based chemotherapy and checkpoint inhibitors. J Clin Oncol. 2021 Aug 1;39(22):2474-2485.

