(UroToday.com) In an oral abstract presentation on the second day of the American Society for Clinical Oncology (ASCO) Genitourinary Cancer Symposium 2022 focussed on urothelial carcinoma, Dr. Crabb presented results of the rucaparib arm of the ATLANTIS trial among patients with metastatic urothelial carcinoma (mUC). Among patients with mUC, a subset of approximately 25%have a DNA repair deficiency (DRD) phenotype which is associated with response to platinum-based chemotherapy (PBC). Given this mechanism of effect, the authors postulated that switch maintenance with the poly(ADP-ribose) polymerase (PARP) inhibitor rucaparib would improve outcomes for patients who had derived benefit from PBC.
This question was assessed within the context of the adaptive, multi-comparison, phase II ATLANTIS trial platform.
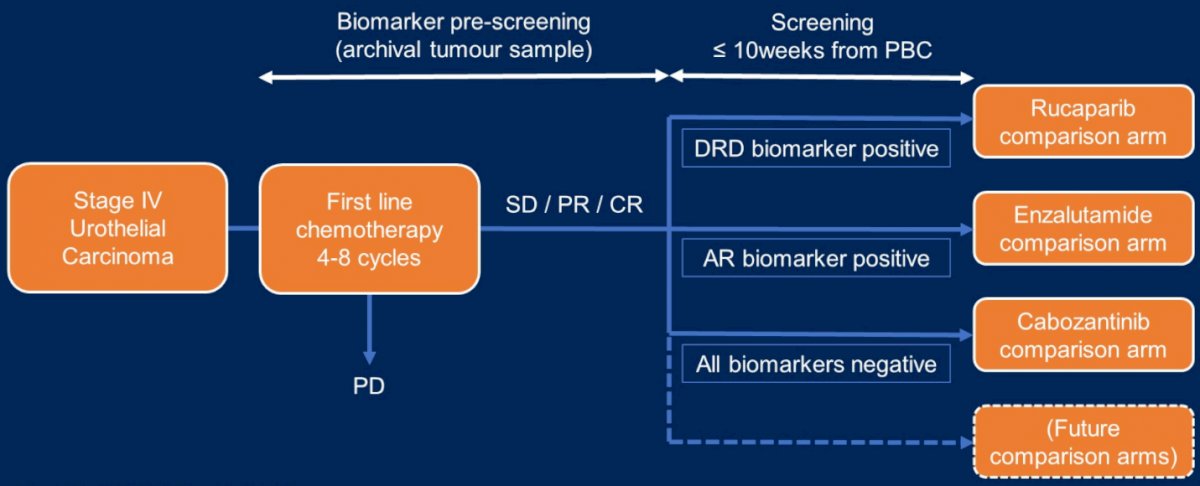
This platform trial tests multiple biomarker selected maintenance therapies for mUC among patients who did not have disease progression after 4 to 8 cycles of PBC. Biomarker-driven allocation of maintenance therapies followed this initial chemotherapy period. Allocation to the rucaparib comparison was based on ≥10% genomic loss of heterozygosity (%LOH) and/or somatic alteration in defined DRD associated genes including ATM, BARD1, BRCA1, BRCA2, BRIP1, CDK12, CHEK2, FANCA, NBN, PALB2, RAD51, RAD51B, RAD51C, RAD51D, RAD54L and/or germline BRCA1 or BRCA2 alteration.
DRD biomarker positive patients were randomised (1:1) to maintenance rucaparib 600 mg BID PO, or matched placebo, within 10 weeks of completing PBC, until disease progression.

The primary endpoint was progression-free survival (PFS). Statistical analysis (with a data cut-off of November 17, 2021) was pre-planned to target a hazard ratio of 0.5 using a 20% 1-sided alpha for this signal-seeking phase II trial with 85.4% power. This required 30 PFS events in 40 patients. Recruitment was stopped after 40 patients due to a combination of the COVID-19 pandemic and the approval of avelumab in the maintenance setting.
Progression-free survival (PFS) using RECIST 1.1 was compared between trial arms, by intention to treat, within a Cox model incorporating baseline minimization factors. Adverse events (AE) were assessed by CTCAE v4.03.
Among 279 patients registered to ATLANTIS and 248 patients pre-screened for the rucaparib comparison, 74 (26.5%) patients were biomarker positive. 40 were randomized within the rucaparib comparison between December 2017 and December 2020. Patients met biomarker positivity by ≥10% loss of heterozygosity (n=22, 55%), DRD gene alteration (n=11, 27.5%), or both (n=7, 17.5%).
As expected based on the randomized design, patient characteristics were balanced between treatment arms including a median age of 70.5 years, male predominance (82.5%), and bladder tumor primary (87.5%). Further, just over half (52.5%) of patients had ECOG performance status 0, 62.5% had previously received cisplatin, and 45% had visceral metastases.
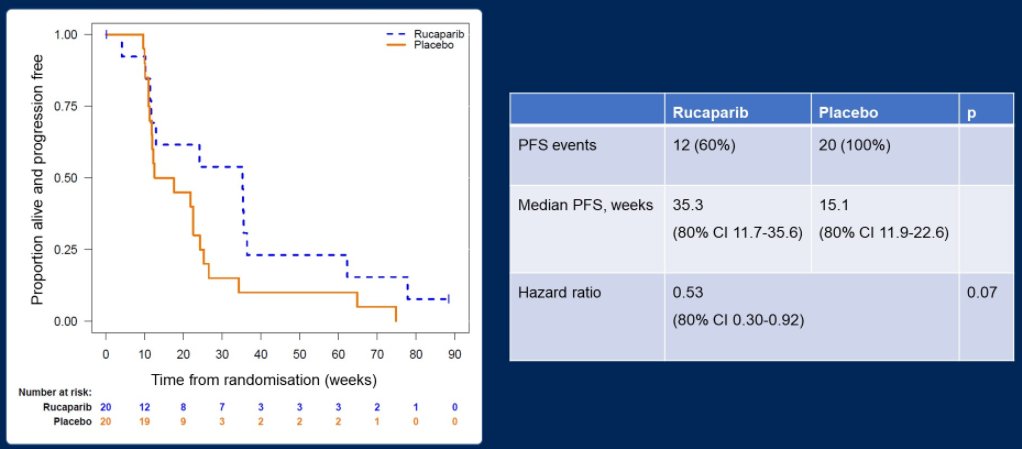
In terms of the primary outcomes of progression-free survival, 12 (60%) and 20 (100%) PFS events occurred in the rucaparib and placebo arms, respectively, over a median follow-up duration of 94.6 weeks in those still alive. The median PFS was 35.3 weeks (80% confidence interval (CI) 11.7-35.6) in patients receiving rucaparib and 15.1 weeks (80% CI 11.9-22.6) in those receiving placebo (hazard ratio 0.53, 80% CI 0.30-0.92, 1 sided p = 0.07).
In terms of overall survival (a key secondary endpoint), the median was not reached among those receiving rucaparib while it was 72.3 weeks (80% CI 51.6-85.4) in those receiving placebo (hazard ratio 1.22, 80% CI 0.62-2.38, p=0.35). There was one partial response among patients treated with rucaparib.
Rucaparib was tolerable with a median duration of 10 rucaparib or 6 placebo cycles on treatment. In the safety population (n = 39), treatment-related adverse events were mostly low grade. The most frequent treatment-related AEs (all grades) of fatigue (63.2% vs 30.0%, p = 0.03), nausea (36.9% vs 5.0%, p = 0.03) and rash (21.1% vs 0%, p = 0.04) were more common with rucaparib respectively. In terms of grade 3 or greater events, one fatigue event, one anemia event, and one hypertension event were observed in patients treated with rucaparib and one patient treated with placebo had grade 3 or greater elevated ALP levels.
In conclusion, Dr. Crabb heightened that maintenance rucaparib was tolerable and extended progression-free survival among DRD biomarker-positive patients with mUC following initial platinum-based chemotherapy.
Presented by: Simon J. Crabb, PhD, MBBS, Southampton Experimental Cancer Medicine Centre, Southampton, United Kingdom

