(UroToday.com) The 2022 GU ASCO Annual meeting included a potpourri of hot topics in urothelial carcinoma session featuring a presentation by Dr. Janet Kukreja discussing clinical implications of molecular biomarkers in upper tract urothelial carcinoma. Dr. Kukreja started by highlighting what a good upper tract urothelial carcinoma biomarker may be able to tell us. First, is there cancer? Second, is it high grade or low grade disease? Currently, there is biopsy discordance with nephroureterectomy specimens. Third, are targeted treatments an option? And finally, is the tumor PDL1/PD1 high or low?
With regards to whether there is cancer in the upper tract, there are several urothelial carcinoma biomarkers on the market including CxBladder, AssureMDx, BTA, BTA Stat (qualitative), NMP22 (qualitative), Survivin (enzyme-line immunosorbent assay), UroVysion, Immunocyt, Bladder EpiCheck, and urine cytology. For assessing the upper tract, Bladder EpiCheck and urine cytology are done by ureteral specific direct catheterization to obtain samples. Among specimens for 82 patients collected between 2018-2020, Pierconti et al.1 reported a 97.4% sensitivity for high grade disease for the Bladder EpiCheck test versus 59% for urine cytology.
Ultimately, Dr. Kukreja wonders if it is possible to translate bladder biomarkers to upper tract disease. Yang et al.2 prospectively sequenced 118 tumors and matched blood DNA from Chinese patients with urothelial carcinoma using next-generation sequencing techniques, including 45 upper tract urothelial carcinomas and 73 bladder urothelial carcinomas. Additionally, 226 patients with upper tract urothelial carcinoma and 350 patients with bladder urothelial carcinoma were assessed from The Cancer Genome Atlas. This study found that there were marked disparities in the mutational landscape for urothelial carcinoma according to site of origin. Signature 22 for exposure to aristolochic acid was only observed in the upper tract urothelial carcinoma cohort. Conversely, signature 6 for defective DNA mismatch repair only existed in the bladder urothelial carcinoma cohort. Compared with bladder urothelial carcinoma, upper tract urothelial carcinoma had higher clonal and subclonal mutation numbers:
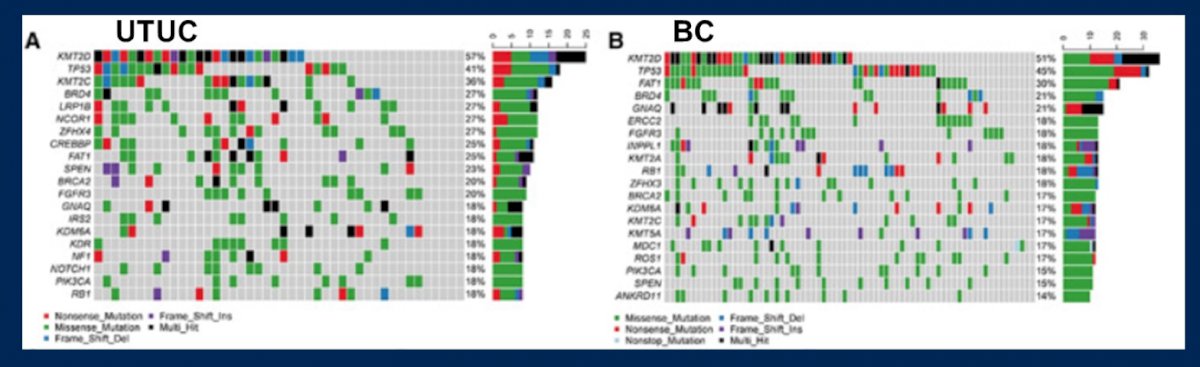
Dr. Kukreja notes that there are several upper tract urothelial carcinoma alterations that biomarkers should include, for example, FGFR3 (for low grade?), HRAS (for high grade?), KMT2D (for ureteral vs pelvic location?), TP53 (for high grade?), mismatch repair, and MSI high status. Specific to FGFR, FGFR3 is more common in upper tract disease than the bladder, which may be a potential target for erdafitinib, as well as infigratinib (which has been assessed in cholangiocarcinoma), rogaratinib, and AZD4547.
Trials assessing PDL1/PD1 directed adjuvant therapy for urothelial carcinoma did include patients with upper tract urothelial carcinoma, but the sample size for these patients is much smaller than for bladder urothelial carcinoma. In IMvigor-010 assessing adjuvant atezolizumab3 showed a median OS of 25.8 months compared to 15.8 months for those undergoing observation. In CheckMate 274 assessing adjuvant nivolumab,4 there was no benefit for those with upper tract disease for the endpoint of recurrence-free survival. The results of the AMBASSADOR trial may shed additional light on this disease space, but these results are still pending.
Dr. Kukreja concluded her presentation of clinical implications of molecular biomarkers in upper tract urothelial carcinoma with the following take-home messages:
- Upper tract urothelial carcinoma is genomically different than bladder urothelial carcinoma
- As such, biomarkers for upper tract disease will accordingly be different
- Currently, the ideal marker does not exist, but we are on our way with more personalized medicine
Presented by: Janet B. Kukreja, MD, University of Colorado, Aurora, CO
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022
References:
- Pierconti F Martini M, Fiorentino V, et al. Upper urothelial tract high-grade carcinoma: Comparison of urine cytology and DNA methylation analysis in urinary samples. Hum Pathol 2021 Dec;118:42-48.
- Yang K, Yu W, Liu H, et al. Comparison of genomic characterization in upper tract urothelial carcinoma and urothelial carcinoma of the bladder. Oncologist. 2021 Aug;26(8):e1395-e1405.
- Bellmunt J, Hussain M, Gschwend JE, et al. Adjuvant atezolizumab versus observation in muscle-invasive urothelial carcinoma (IMvigor010): A multicentre, open-label, randomized, phase 3 trial. Lancet Oncol. 2021 Apr;22(4):525-537.
- Bajorin DF, Witjes JA, Gschwend JE, et al. Adjuvant nivolumab versus placebo in muscle-invasive urothelial carcinoma. N Engl J Med. 2021 Jun 3;384(22):2102-2114.

