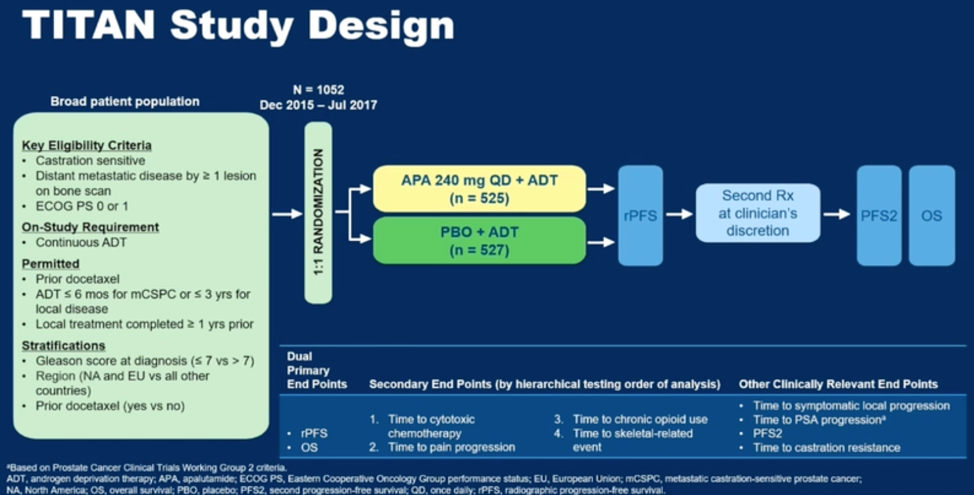
In the initial analysis presented at 22.7 months of follow-up, treatment with apalutamide resulted in a statistically significant rPFS and overall survival benefit as illustrated with the Kaplan-Meier curves below (HR 0.48 for rPFS or death, HR 0.67 for death)
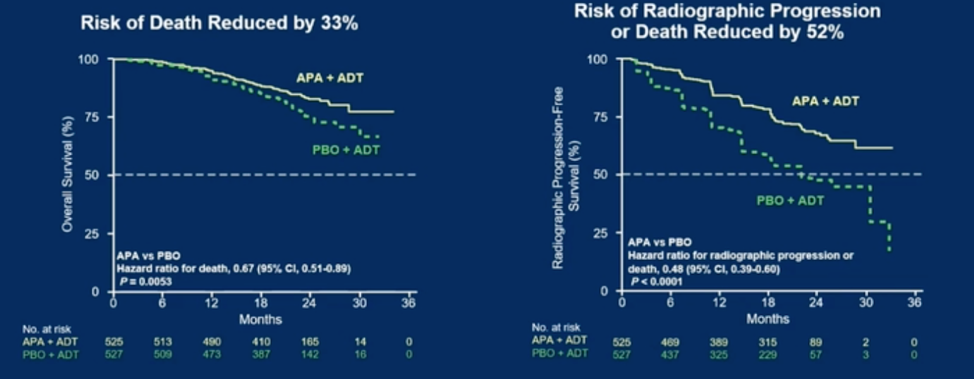
In this presentation, Dr. Chi presented follow-up data from a median of 44 months of clinical follow-up. This included an intention to treat analysis of all patients, though 40% of the placebo group crossed over from placebo to apalutamide after the initial unblinding at 22.7 months of follow-up. In this analysis, the benefit of apalutamide on overall survival was confirmed, with a similar hazard ratio for death – HR 0.65, 95% CI 0.53 – 0.79, p < 0.001. The median overall survival in the placebo group was 52.2 months, and the median overall survival in the apalutamide group has not yet been reached.

The benefit of apalutamide held after a sensitivity analysis using a pre-specified inverse probability of censoring weighting method. Other endpoints such as second progression-free survival (median not reached vs 44 months in placebo), time to castration resistance (median not reached vs 11.4 months for placebo) also favored apalutamide. Subgroup analysis suggested benefit for apalutamide in almost all subgroups including both low and high-volume metastatic disease. There was a trend towards favoring placebo in patients who had received prior docetaxel, but Dr. Chi noted that this was only 10% of the trial population and a post-hoc interaction test showed not interaction between the efficacy of apalutamide and prior docetaxel.
Finally, patient report quality of life data (FACT-P total score) and cumulative incidence of side effects were roughly similar, including in the domains of falls, fatigue, fractures and serious adverse events. Apalutamide-treated patients had a higher incidence of rashes, which plateaued at just under 40% by 60 months.
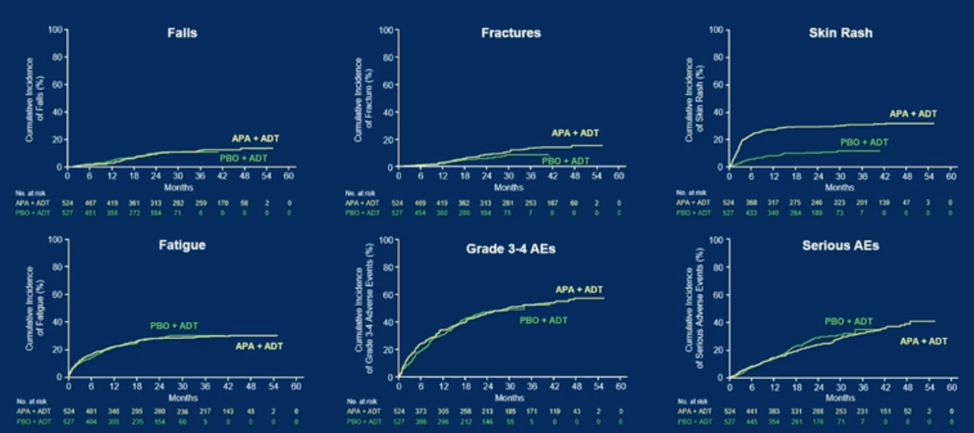
Dr. Chi concluded that after approximately four years of follow-up, data confirm the survival benefit over apalutamide for men with mCSPC relative to placebo as well as in other endpoints such as time to castration resistance and was not associated with marked differences in key adverse events like falls or fatigue.
Presented by: Kim Chi, MD, FRCP(C), medical oncologist, a senior research scientist, professor of medicine at the University of British Columbia and recently named the head of BC Cancer. He is also the Co-Chair of the Genitourinary Disease Site for CCTG
References:
1. Chi K, Agarwal N, Bjartel A, et al., Apalutamide for Metastatic, Castration-Sensitive Prostate Cancer. N Engl J Med 381, (2019):13-24

