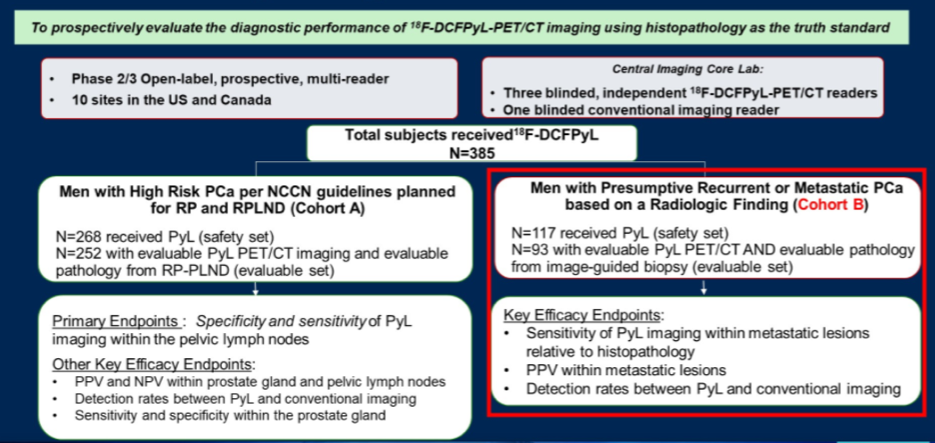
The authors included 117 men with radiologic evidence of local recurrence or metastatic disease on baseline anatomical imaging (CT, MRI) or whole-body bone scintigraphy and in whom at least one lesion was deemed amenable to biopsy. 18F-DCFPyL-PET/CT was performed using a single dose of 9 mCi (333 MBq) of 18F-DCFPyL, administered via intravenous injection, and followed by PET/CT acquisition 1 to 2 hours thereafter. 18F-DCFPyL-PET/CT detection rates including lesion counts were systematically analyzed according to TNM staging: prostatic (T), pelvic LN (N), extra-pelvic LN (M1a), bone (M1b), and other visceral organs/soft tissue (M1c) with three central, blinded, and independent readers evaluating the 18F-DCFPyL scans.
The authors included 82 (70%) patients who had baseline radiographic M1 stage disease (14 patients with M1a, 50 patients with M1b, 18 patients with M1c), and 33 (28%) patients who had M0 disease based on conventional imaging (with central review), and two patients were unevaluable.
In patients with M0 disease based on conventional imaging, 18F-DCFPyL-PET/CT up-staged 19 of 33 patients (58%) to M1 disease, of whom 91% (10/11) who underwent an extra-pelvic biopsy were confirmed to have M1 disease by pathology, including 9 patients with M1b and 1 patient with M1a.
In patients with M1 disease based on conventional imaging, 18F-DCFPyL-PET/CT upstaged 10 of 64 (16%) and down-staged 18 of 82 (22%) to M0 disease. Among those who were upstaged, 4 patients were upstaged from M1a to M1b/c disease and 6 patients were upstaged from M1b to M1c disease.

On the basis of these data, the authors conclude that 18F-DCFPyL-PET/CT may be a useful tool in staging men with both metastatic and nonmetastatic relapsed prostate cancer.
Presented by: Jeremy C. Durack, MD, MS, Interventional Radiologist, Memorial Sloan Kettering Cancer Center
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter during the 2021 ASCO Genitourinary Cancers Symposium (ASCO GU), February 11th to 13th, 2021

