ARCHES randomized 1,150 mHSPC patients 1:1 to receive enzalutamide (160 mg/day) plus ADT or placebo plus ADT, stratified by disease volume and prior docetaxel use. In this post hoc analysis, patients previously centrally categorized as MX/unknown metastasis at initial diagnosis (n=213) were adjudicated as either M0 or M1 disease following review of their medical profiles (n=77 to M0; n=123 to M1; n=13 remained MX). Subsequently, efficacy outcomes, include radiographic progression-free survival and key secondary endpoints (time to PSA progression, time to initiation of new antineoplastic therapy, PSA undetectable rate, and objective response rate) were compared according to M0 versus M1 disease at initial diagnosis.
In this analysis, 246 patients (enzalutamide n=117; placebo n=129) had M0 and 890 patients (enzalutamide n=448; placebo n=442) had M1 disease at initial diagnosis. Baseline characteristics were generally comparable between treatment arms, however a greater proportion of M1 patients had high volume disease (n=606; 68.1% vs n=116; 47.2% [M0]) and prior docetaxel (n=173, 19.4% vs n=29, 11.8% [M0]).
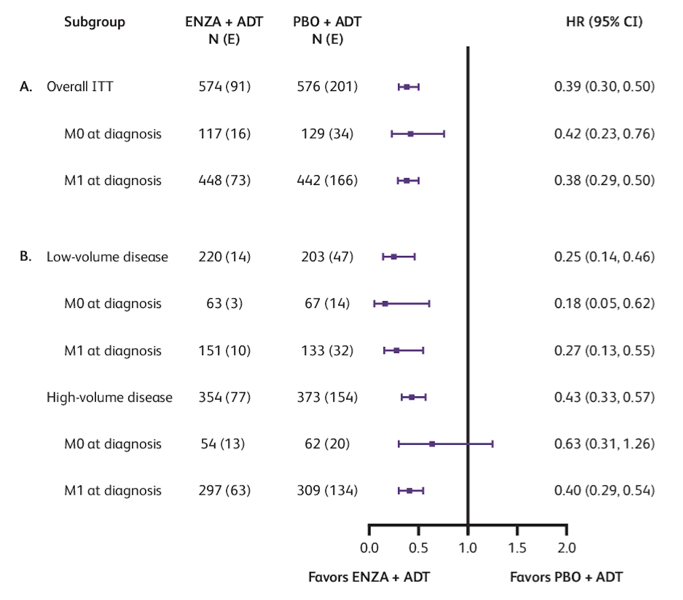
Enzalutamide plus ADT improved radiographic progression-free survival versus placebo plus ADT in both M0 at diagnosis (HR 0.42, 95% CI 0.23-0.76) and M1 at diagnosis (HR 0.38, 95% CI 0.29-0.50) subgroups, which is consistent with the overall ITT population (HR 0.39, 95% CI 0.30-0.50). Additionally, radiographic progression-free survival results favored enzalutamide plus ADT in patients grouped according to a combination of disease volume at baseline and metastatic disease status at diagnosis:
For both M0 and M1 subgroups, median radiographic progression-free survival was not reached with enzalutamide plus ADT, versus 22.1 and 16.6 months, respectively, with placebo plus ADT:

Similar improvements for enzalutamide plus ADT versus placebo plus ADT were observed for secondary endpoints:
- Time to PSA progression: M0 HR 0.12, 95% CI 0.04-0.34; M1 HR 0.20, 95% CI 0.14-0.28
- Time to new antineoplastic therapy: M0 HR 0.32, 95% CI 0.15-0.66; M1 HR 0.27, 95% CI 0.18-0.40
- PSA undetectable rate (% difference): M0 45.4, 95% CI 33.9-56.9; M1 53.6, 95% CI 48.1-59.1
- Objective response rate (% difference): M0 18.5, 95% CI -7.4 to 44.4; M1 20.1, 95% CI 10.9 to 29.4
Dr. Azad concluded this presentation of a post hoc analysis of ARCHES with the following summary points:
- There is clinical benefit of enzalutamide plus ADT compared to placebo plus ADT based on radiographic progression-free survival and secondary endpoints in men who progressed from M0 to M1 HSPC and those with de novoM1 HSPC
- These findings will aid to inform clinical decision-making regarding the use of enzalutamide in patients with mHSPC in these subgroups
Co-Authors: Arnauld Villers, Boris Alekseev, Russell Zelig Szmulewitz, Antonio Alcaraz, Neal D. Shore, Daniel Peter Petrylak, Jeffrey Holzbeierlein, Francisco Gomez-Veiga, Brad Rosbrook, Fabian Zohren, Ho-Jin Lee, Gabriel P. Haas, Taro Iguchi, Arnulf Stenzl, Andrew J. Armstrong; Monash Health, Melbourne, Victoria, Australia; University Hospital Centre, Lille University, Lille, France; Hertzen Moscow Cancer Research Institute, Moscow, Russian Federation; The University of Chicago, Chicago, IL; Hospital Clinic de Barcelona, Barcelona, Spain; Carolina Urologic Research Center, Myrtle Beach, SC; Yale Cancer Center, New Haven, CT; University of Kansas Medical Center, Kansas City, KS; Hospital Universitario de Salamanca, GITUR-IBSAL, Salamanca, Spain; Pfizer Inc., San Diego, CA; Astellas Pharma Inc., Northbrook; Osaka City University Graduate School of Medicine, Osaka, Japan; University Hospital, Eberhard Karls University, Tübingen, Germany; Duke Cancer Institute Center for Prostate and Urologic Cancers, Durham, NC
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md during the 2021 ASCO Genitourinary Cancers Symposium (ASCO GU), February 11th to 13th, 2021
References:

