At ESMO 2020, Dr. Choueiri presented the first results from the CheckMate-9ER of the combination of nivolumab and cabozantinib, as compared to sunitinib, in the first-line treatment of patients with advanced or metastatic renal cell carcinoma. This first report, which is still unpublished, demonstrated the superiority of nivolumab plus cabozantinib to sunitinib in the first-line treatment of patients with advanced renal cell carcinoma with respect to both progression-free survival and overall survival and led to its approval. In a plenary abstract presentation in the Poster Highlights Session: Renal Cell Cancer - Clinical Trial Updates session at the 2021 ASCO GU Cancers Symposium, Dr. Robert Motzer presented updated results of CheckMate-9ER assessing the durability of benefit, as well as assessing outcomes among patients with sarcomatoid features, an aggressive histologic subtype associated with poor prognoses.
While previously reported, to briefly summarize, the phase III, open-label CheckMate 9ER trial (NCT03141177) randomized patients with advanced RCC in a 1:1 fashion to receive nivolumab 240 mg IV Q2W + cabozantinib 40 mg PO QD (N+C; n = 323) or sunitinib (S) 50 mg PO (4 weeks of 6-week cycles; n = 328) in the first-line setting, with randomization stratified by International Metastatic Renal Cell Carcinoma Database Consortium risk score, tumor programmed death ligand 1 expression, geographic region. Treatment was continued until disease progression or unacceptable toxicity. The primary endpoint was RECIST v1.1-defined progression-free survival (PFS) by blinded independent central review (BICR) in all randomized (intent-to-treat [ITT]) patients. Important secondary endpoints included overall survival (OS), objective response rates (ORR) by BICR, and safety. Further, patients with and without sarcomatoid RCC were identified by local pathology reports, and outcomes in these patients were evaluated via prespecified subset analyses.

Among 651 patients in the ITT population, sarcomatoid histology was identified in 75 patients (11.5%), not identified in 557 (84.6%), and was unavailable in 19 (2.9%). Among these 75 patients, 34 were randomized to nivolumab and cabozantinib and 41 were randomized to sunitinib.
In contrast with the primary analysis with a database lock as of March 30, 2020, the present analysis provides updated results at 18.1 months of follow-up as of a database lock of September 10, 2020.
With these updated data, the authors demonstrate continued significant improvements in both progression-free survival (hazard ratio 0.52, 95% confidence interval 0.43-0.64) and overall survival (hazard ratio 0.66, 95% confidence interval 0.50-0.87).

Among those with sarcomatoid histology, patients treated with nivolumab and cabozantinib had significantly improved progression-free survival, overall survival, and objective response rates, compared with sunitinib. Further, this relative benefit, compared to sunitinib, was even larger in patients with sarcomatoid histology compared to those without sarcomatoid histology.
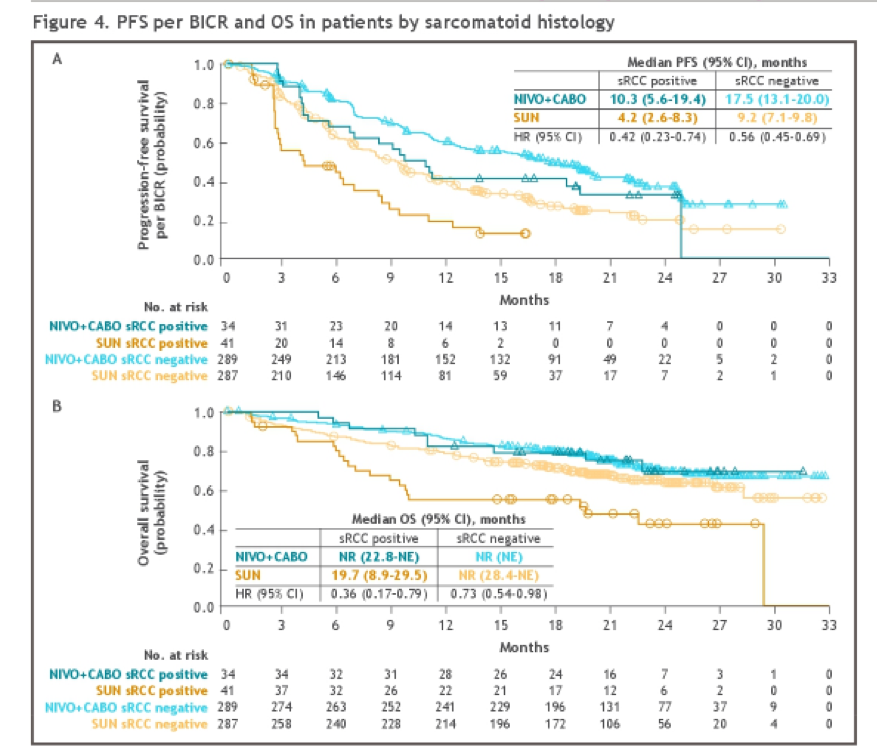
The authors, therefore, conclude that nivolumab and cabozantinib demonstrated improved efficacy and survival outcomes, compared to sunitinib, regardless of the presence of sarcomatoid histology.
Presented by: Robert J. Motzer, MD, Medical Oncologist, Memorial Sloan Kettering Cancer Center
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter during the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (#GU21), February 11th-February 13th, 2021

