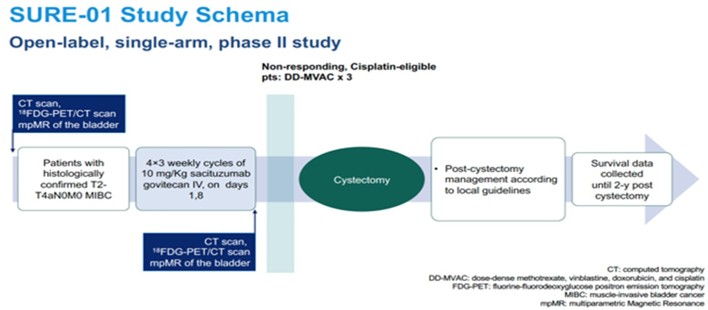
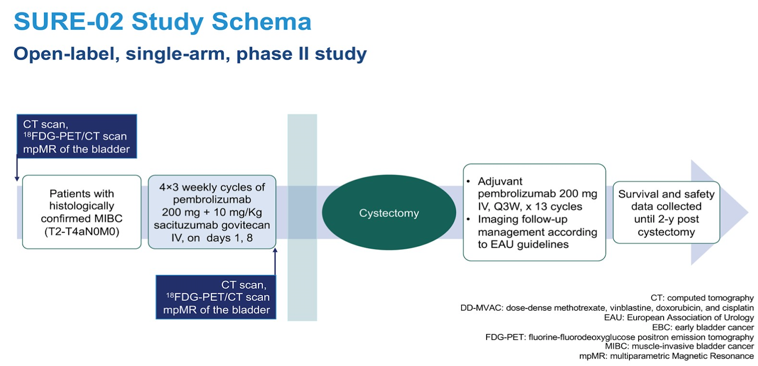
The SURE trial is a phase II, open-label trial that will test the safety, tolerability, and activity, of sacituzumab govitecan and sacituzumab govitecan plus pembrolizumab. This study will enroll patients sequentially in the two cohorts. Patients should have a histopathologically-confirmed predominant urothelial carcinoma, be fit and planned for radical cystectomy, have clinical stage T2-T4N0M0 muscle-invasive bladder cancer, be ineligible (Galsky criteria), or refuse to receive cisplatin-based chemotherapy. Eligible patients will receive 4 cycles of 10 mg/Kg sacituzumab govitecan IV, on days 1, 8, of each 21-day cycle (SURE-01) and sacituzumab govitecan plus pembrolizumab on day 1, every 21 days, at the standard dose of 200 mg intravenously (SURE-02). Surgery will be planned at the time of study inclusion to be performed within 2 weeks of the last dose of study drug. After surgery patients will be managed and the surgical safety data will be recorded according to the European Association of Urology guidelines. In SURE-02, an adjuvant phase of 13 postoperative cycles of pembrolizumab will be administered.
The SURE-01 study schema is as follows:
As follows is the SURE-02 trial design:
The primary endpoint of the study is to assess the proportion of ypT0N0. Secondary endpoints will be to evaluate radiological response for those patients with measurable disease, evaluate the surgical and medical safety of neoadjuvant therapy, and assess survival outcomes (event-free survival and overall survival). The total sample size of SURE is of 77 patients, distributed as 56 patients in SURE-01 and 48 in SURE-02. The assumptions include a ypT0N0 ≤20% as H0 and ≥45% as H1 in a single-stage A’Hern’s design for SURE-01 and a 2-stage design for SURE-02 assuming a ypT0N0 ≤30% as H0 and ≥45% as H1. In SURE-02 a safety lead-in phase will be conducted including 10 patients. An external review committee will evaluate the safety outcomes in this phase and the occurrence of pre-defined study-limiting events. Biomarker analyses will include assessment of transcriptomic clustering, immune-gene signature, next-generation sequencing on tumor circulating tumor DNA, including single-cell RNA sequencing on frozen tumor samples, before and after treatment.
Clinical trial information: 2020-004844-27.
Presented by: Andrea Necchi, MD, Medical Oncologist, Department of Medical Oncology, Fondazione IRCCS - Istituto Nazionale dei Tumori, Milan, Italy
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md during the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (#GU21), February 11th-February 13th, 2021
References:

