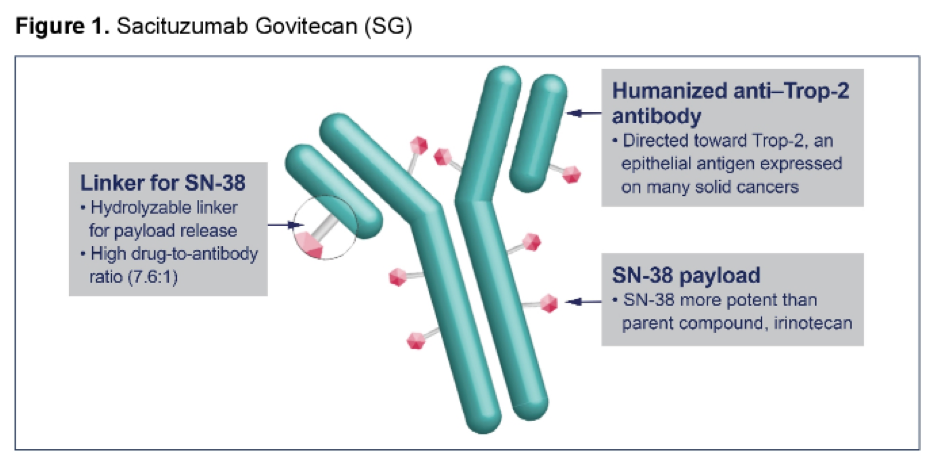
The TROPHY-U-01 study, a phase II registrational study, demonstrated an objective response rate (ORR) of 27% in 113 patients receiving sacituzumab govitecan for advanced urothelial carcinoma following prior platinum-based and CPI therapies (median 3 prior lines of therapy and 87% with ≥1 Bellmunt risk factors) in data presented at ESMO 2020. Median overall survival (OS) in this population was 10.5 months. Both ORR and OS in this cohort exceeded that expected on the basis of historic single-agent chemotherapy with ORR and OS of less than 10%; and 7 months, respectively.
In a presentation at the 2021 ASCO GU Cancers Symposium, Dr. Grivas described the design of the confirmatory phase III trial, TROPiCS-04 (NCT04527991). TROPiCS-04 is a global, multicenter, open-label, randomized controlled trial, performed among patients with locally advanced unresectable or metastatic urothelial carcinoma. To be eligible for inclusion, patients much have had disease progression following prior platinum-based and CPI therapies. Specific inclusion criteria require Eastern Cooperative Oncology Group performance status 0–1 and adequate hematologic, hepatic, and renal function.

In the randomized design, patients will be allocated in a 1:1 fashion to receive sacituzumab govitecan 10 mg/kg intravenously (IV) on day 1 and 8 of 21-day cycles or physician’s choice single agent chemotherapy (paclitaxel 175 mg/m2, docetaxel 75 mg/m2, or vinflunine 320 mg/m2 IV on day 1 of 21-day cycles). Patients will continue on their assigned treatment until progressive disease, unacceptable toxicity, or withdrawal of consent. For those who derived clinical benefit, treatment beyond progressive disease will be permitted.
The authors aim to enroll approximately 482 patients to provide 90% power on the primary endpoint of overall survival, with key secondary endpoints assessed including progression-free survival, ORR, clinical benefit rate, duration of response (all per Response Evaluation Criteria in Solid Tumors v1.1), safety, and quality of life.
Presented by: Petros Grivas, MD, Ph.D., Associate Professor, Clinical Director of Genitourinary Cancers Program, University of Washington, Associate Member, Clinical Research Division, Fred Hutchinson Cancer Research Center.
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter during the 2021 ASCO Genitourinary Cancers Symposium (ASCO GU), February 11th to 13th, 2021
Related Content:
Genitourinary Cancers Symposium 2021 Trials in Progress TROPiCS-04 - Petros Grivas

