Summary
This abstract provides data from a phase III randomized trial in patients with high risk urothelial carcinoma. Patients were randomized to receive either postoperative radiotherapy (PORT) or PORT plus chemotherapy (cisplatin and gemcitabine). Chemotherapy was split up to two cycles before PORT and two cycles after PORT. Patients must have had at least one high risk feature including ≥pT3b, grade 3, or positive nodes with negative margins after radical cystectomy. The radiation protocol was 45Gy in 1.5Gy BID.
In this study, 81 patients with urothelial carcinoma were enrolled.

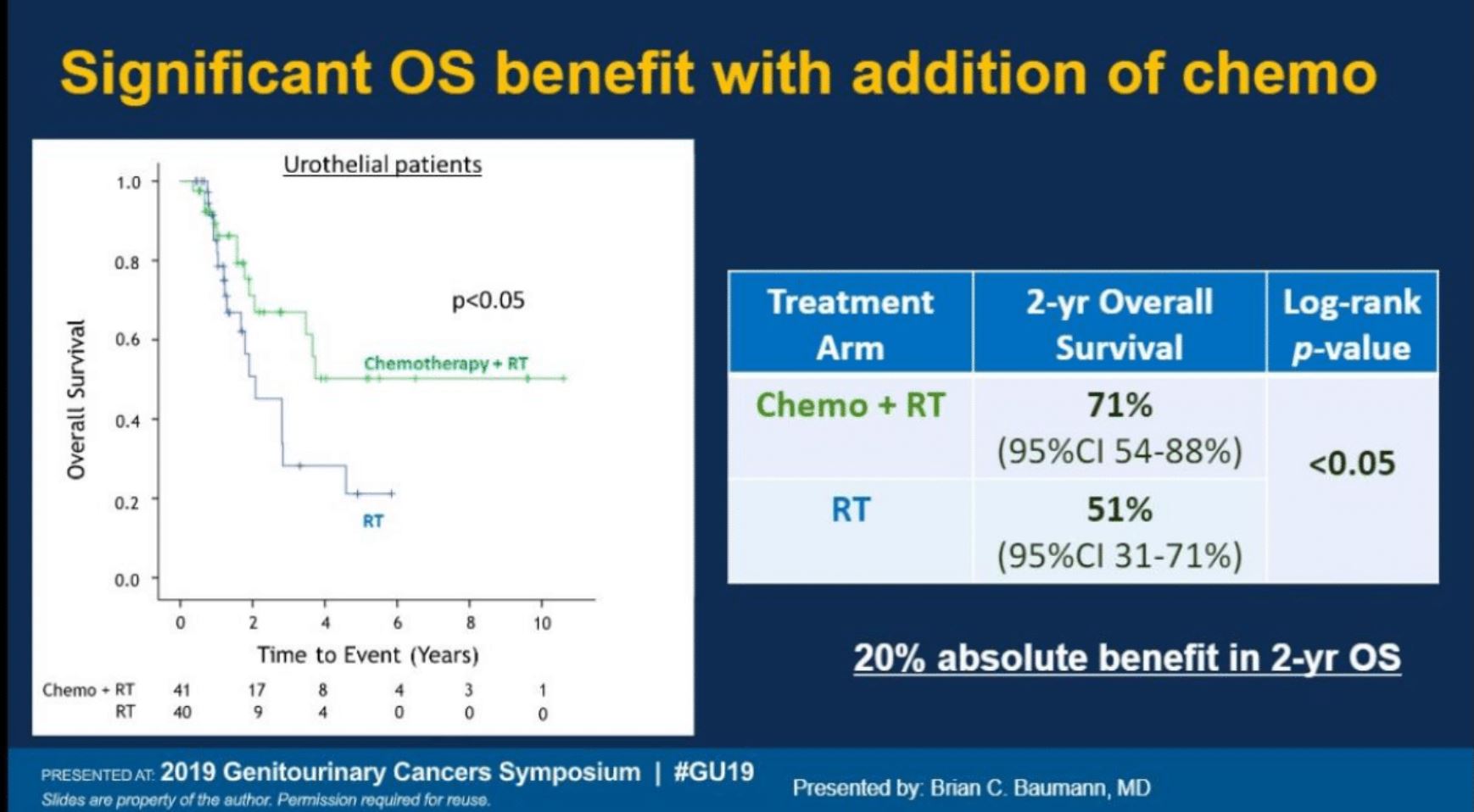
This was a young cohort of patients with a median age of 55. This trial started before neoadjuvant chemotherapy was routinely given for muscle-invasive bladder cancer in Egypt. With 21-month median follow up time, the two year disease free survival was 62% for patients receiving PORT plus chemotherapy compared with 48% receiving PORT alone. PORT plus chemotherapy also improved overall survival at two years, 71% vs 51% (HR 0.45, 95% CI 0.21-0.96, p=0.039)
In terms of safety, grade III GI toxicity was observed in 7% of patients receiving chemotherapy plus PORT compared to 8% of PORT patients.
Conclusions
For patients who did not receive neoadjuvant chemotherapy, PORT plus chemotherapy improves overall survival compared with PORT alone, with an impressive hazard ratio of 0.45. Interestingly, for patients with non-urothelial bladder cancer, Dr. Zaghloul mentioned that adjuvant chemoradiation was detrimental. Adjuvant chemoradiation in this cohort was well tolerated and contemporary studies should be conducted to confirm the benefit seen here.
Presented by: Mohamed S. Zaghloul, MD, Egyptian National Cancer Institute, Cairo, Egypt
Written by: Jason Zhu, MD, Fellow, Division of Hematology and Oncology, Duke University. Twitter: @TheRealJasonZhu at the 2019 American Society of Clinical Oncology Genitourinary Cancers Symposium, (ASCO GU) #GU19, February 14-16, 2019 - San Francisco, CA
References:
1. Stein JP, Lieskovsky G, Cote R, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. Journal of clinical oncology 2001;19:666-75.
2. Leow JJ, Martin-Doyle W, Rajagopal PS, et al. Adjuvant chemotherapy for invasive bladder cancer: a 2013 updated systematic review and meta-analysis of randomized trials. European urology 2014;66:42-54.
3. Sternberg CN, Skoneczna I, Kerst JM, et al. Immediate versus deferred chemotherapy after radical cystectomy in patients with pT3–pT4 or N+ M0 urothelial carcinoma of the bladder (EORTC 30994): an intergroup, open-label, randomised phase 3 trial. The Lancet Oncology 2015;16:76-86.

