Methods:
This was a randomized phase 3 study, where pts with ≤ 2 prior lines of therapy for mUC who progressed during or following platinum treatment were randomized 1:1 to atezo or chemo (vinflunine, paclitaxel or docetaxel, per physician). The primary endpoint was overall survival (OS), hierarchically compared between treatment arms in PD-L1–selected and ITT pts. Planned exploratory biomarker analyses included tGE (RNA-seq) and TMB (FoundationOne).
Results:
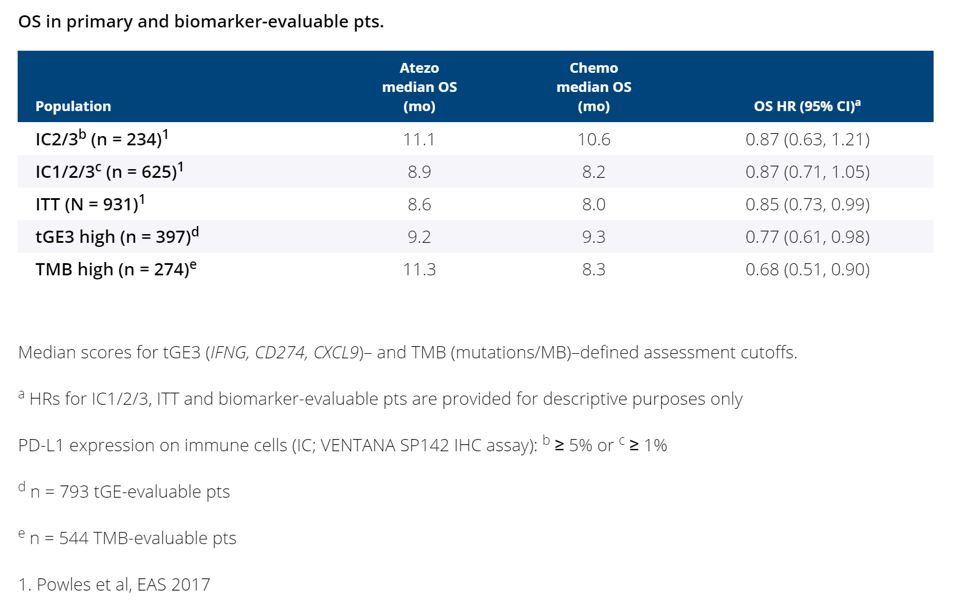
The ITT population included 931 pts (atezo arm, 467; chemo arm, 464), and biomarker-evaluable subgroups were representative of the ITT population. PD-L1 expression positively correlated with tGE (R = 0.61) but not TMB (R = 0.13).
Conclusions:
In this randomized Phase III study, it was demonstrated that high PD-L1 and high tGE are associated with improved outcomes with both chemo and atezo. In contrast, higher TMB predicted OS in favor of atezo; however, clinical benefit with atezo was also seen in the ITT population. Clinical trial information: NCT02302807
Presented by: Thomas Powles, MD Barts Cancer Institute, Queen MARY University of London, UK
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre @GoldbergHanan at the 2018 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, February 8-10, 2018 - San Francisco, CA

