(UroToday.com) The 2023 American Society of Clinical Oncology (ASCO) annual meeting held in Chicago, IL between June 2nd and June 6th was host to a kidney and bladder cancers poster session. Dr. Roberto Pili presented the results of a phase I/II study evaluating the safety, pharmacodynamics, and efficacy of entinostat in combination with atezolizumab and bevacizumab in patients with renal cell carcinoma (RCC).
It has been demonstrated that immunosuppressive modulators such as regulatory T cells (Tregs) and myeloid-derived suppressor cells (MDSCs) limit the efficacy of immunotherapies. By inhibiting the functions of Tregs and MDSCs in preclinical models, histone deacetylase (HDAC) inhibitors have demonstrated antitumor effects in combination with PD-1 inhibition.

The authors hypothesized that HDAC inhibition may enhance or re-sensitize RCC patients to immunotherapy. Accordingly, they conducted a phase I/II clinical trial (NCT03024437) evaluating the HDAC inhibitor entinostat in combination with atezolizumab (immune checkpoint inhibitor) plus bevacizumab (VEGF inhibitor) in metastatic RCC patients who were either treatment naïve or had previously received immune checkpoint inhibitors.
Given the phase 1/2 nature of the study, the primary objective was to evaluate the safety, tolerability, and efficacy of this combination strategy. This study consisted of two phases:
- Phase 1: A dose finding
- Phase 2: Two-stage phase
- Enrollment was halted during Stage 1 of the Phase 2 portion due to “unforeseen circumstances”
In the Phase I dose finding cohorts, patients received oral entinostat, in combination with atezolizumab (1,200 mg/kg IV every three weeks) plus bevacizumab (15 mg/kg IV every 3 weeks), at the following doses, in accordance with the 3+3 standard design:
- 1 mg (dose level 1) orally every 7 days
- 3 mg (dose level 2) orally every 7 days
- 5 mg (dose level 3) orally every 7 days
Phase II patients were split into two distinct cohorts:
- Cohort A (no prior treatments with PD-1 or PD-L1 inhibitors)
- Cohort B (at least one prior treatment with a PD-1 or PD-L1 inhibitor for metastatic disease).
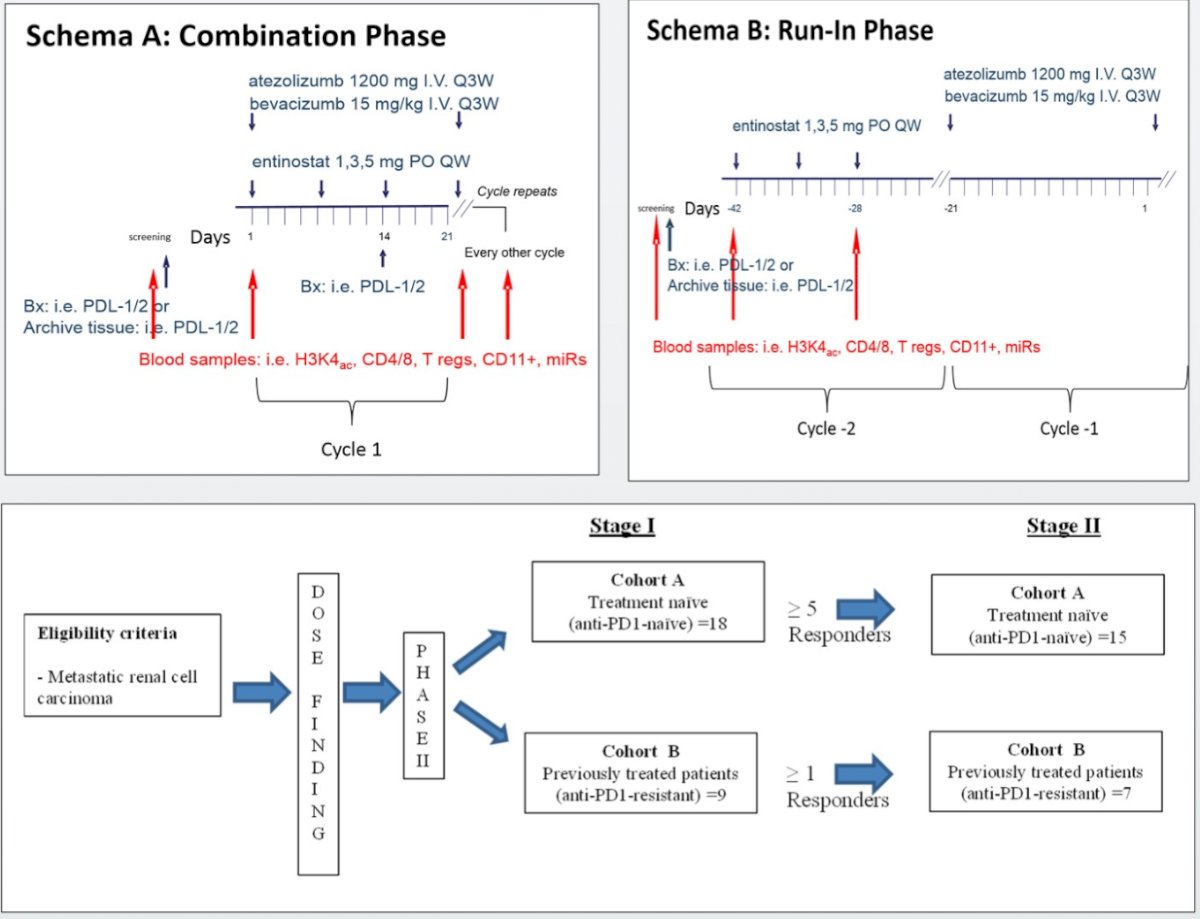
Patients treated at dose level 3 (5 mg) were included in the Phase II efficacy analyses.
This trial enrolled 31 patients (Phase 1: 18 patients; Phase 2: 13 patients).

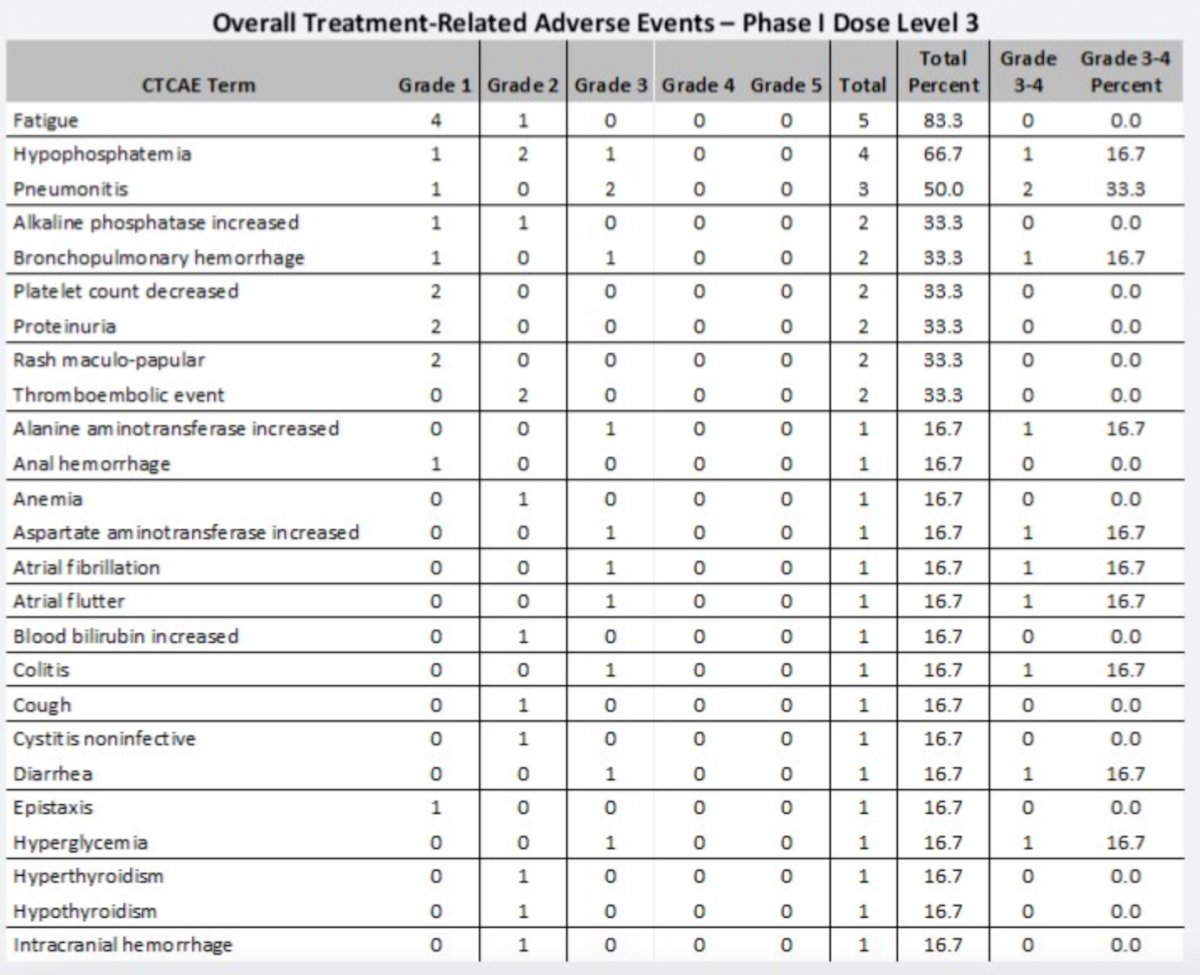
Phase I was completed with the following dose-limiting toxicities:
- Dose level 1: Hypertension (n=3)
- Dose level 2: Encephalopathy and hyponatremia
- Dose level 3: Pneumonitis and pruritis
Dose level 3 was established as the recommended phase 2 dose. A total of 21 grade ≥3 events were observed. The most common treatment-related toxicities were:
- Thrombocytopenia
- Hypophosphatemia
- Proteinuria
- Diarrhea
- Fatigue
- Neutropenia
In the efficacy analyses, treatment responses were as follows:
- Cohort A (no prior PD-1/PD-L1): 9 partial responses for an objective response rate (ORR) of 60% (95% CI: 32.3 – 83.7%)
- Cohort B: 1 partial response for an ORR of 33.3% (95% CI: 28.2 – 56.4%)
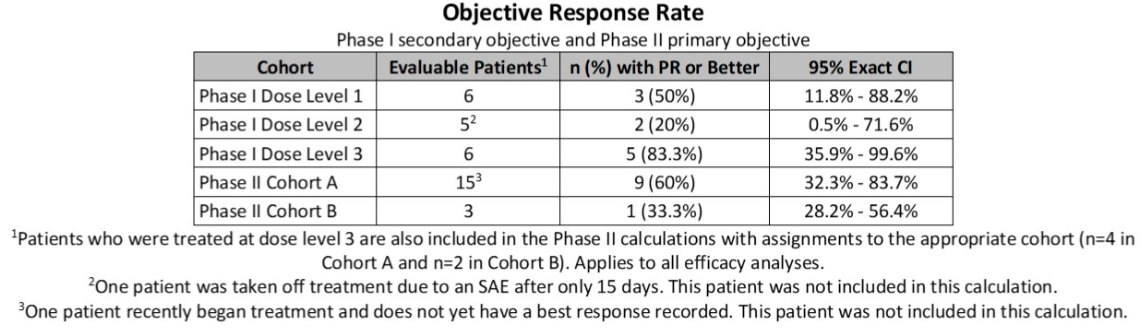
The ORR met the Stage I primary objective per Simon's optimal two-stage design. The median PFS was 13.7 months (95% CI: 8.7 - 21.3) in Cohort A and 9.8 months (95% CI: 1.3 – Not reached) in Cohort B. Dr. Pili did note that the investigators have collected blood samples to conduct correlative studies on peripheral blood mononuclear cells.


Dr. Pili and colleagues concluded that the preliminary results from this study suggest that the combination of entinostat and atezolizumab plus bevacizumab is relatively well-tolerated and may be active in patients with ccRCC.
Presented by: Dr. Roberto Pili, MD, Professor and Chief of the Division of Hematology/Oncology, Department of Medicine, University of Buffalo, Buffalo, NY
Written by: Rashid Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 2023 American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, Fri, June 2 – Tues, June 6, 2023.

