(UroToday.com) Prostate specific membrane antigen (PSMA) is a conserved transmembrane protein present on prostate cells and upregulated following transformation, with long-standing promise as a terrific target for both imaging and therapy. As published a year ago in Lancet1, TheraP (ANZUP 1603) was a phase III randomized study of 177Lu-PSMA-617 versus cabazitaxel in metastatic castration-resistant prostate cancer (mCRPC) following progression on docetaxel. 177Lu-PSMA-617 is a small molecule targeting PSMA and carrying 177Lu, a beta-emitting radionuclide. Here at ASCO 2022, the authors update the previously reported results with overall survival data following a median follow up of three years.
Dr. Hofman briefly reviewed the TheraP study design including key eligibility criteria of chemotherapy-suitable post-docetaxel patients with mCRPC. Adequate organ function and performance status was required (ECOG 0-2). Dual PET CT scans (68Ga-PSMA-11 and 18F-FDG) were performed and qualifying PSMA expression of PSMA SUVmax > 20 (at any site) and SUVmax of at least 10 at all other measurable sites in the absence of a metastatic site with discordant PSMA and 18F detection. Participants were randomized 1:1 to treatment with 177Lu-617 (8.5-6GBq every 6 weeks, maximum 6 cycles) versus cabazitaxel (20 mg/m2 every 3 weeks, maximum 10 cycles). Key results from previous report were reviewed today, included improvement in those patients receiving 177Lu-617 of PSA response rate (66% v 37%), response rate (via RECIST, 49% v 24%), lesser Grade ≥3 toxicities, and patient-reported outcomes, all versus the cabazitaxel arm.
Today, it was described that the PFS curves demonstrated an inconsistent treatment response with respect to time, leading the investigators to reanalyze using restricted mean survival time (RMST). When done, the PFS (both PSA and radiographic) benefit of 177Lu-617 remained, with a HR of 0.62 (95% CI 0.45-0.85, p = 0.0028).

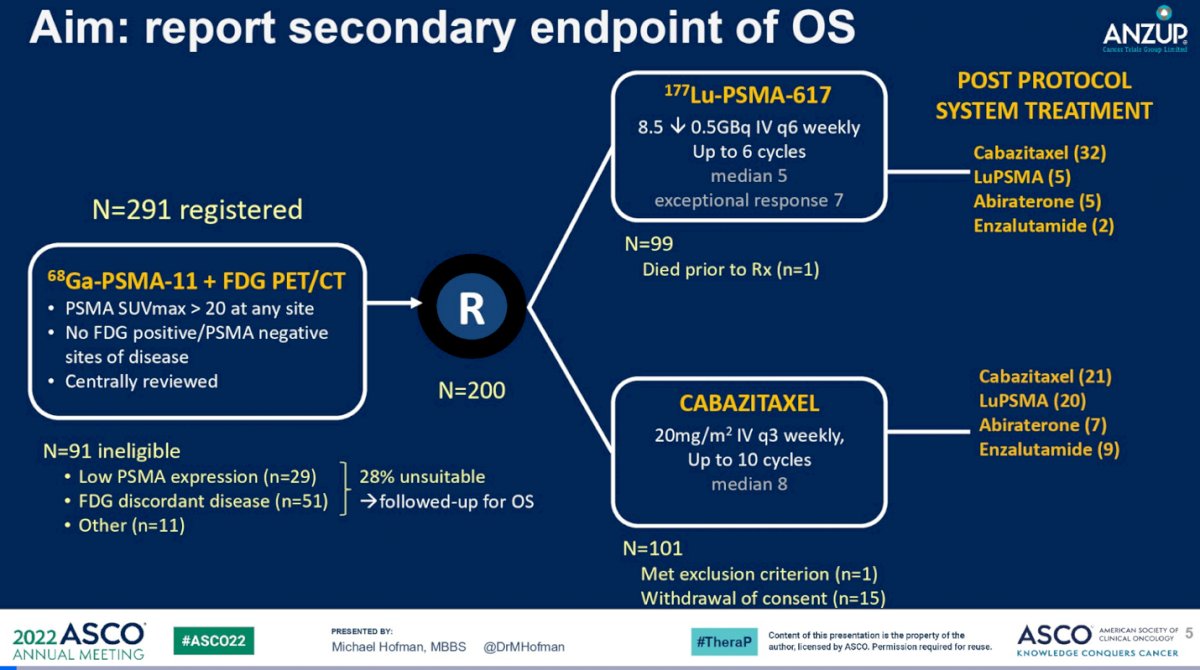
The authors report overall survival (OS) as a key secondary endpoint of the study. OS was analyzed by intention-to-treat and summarized by RMST to account for non-proportional hazards. 291 patients were screened, among which 200 were eligible and randomized. Of the screened patients, 27% (80/291) were excluded to disqualify PET imaging data. 51 has SUVmax of <20 and 29 had discordant imaging. In this presentation, follow up data for these patients was also shared.
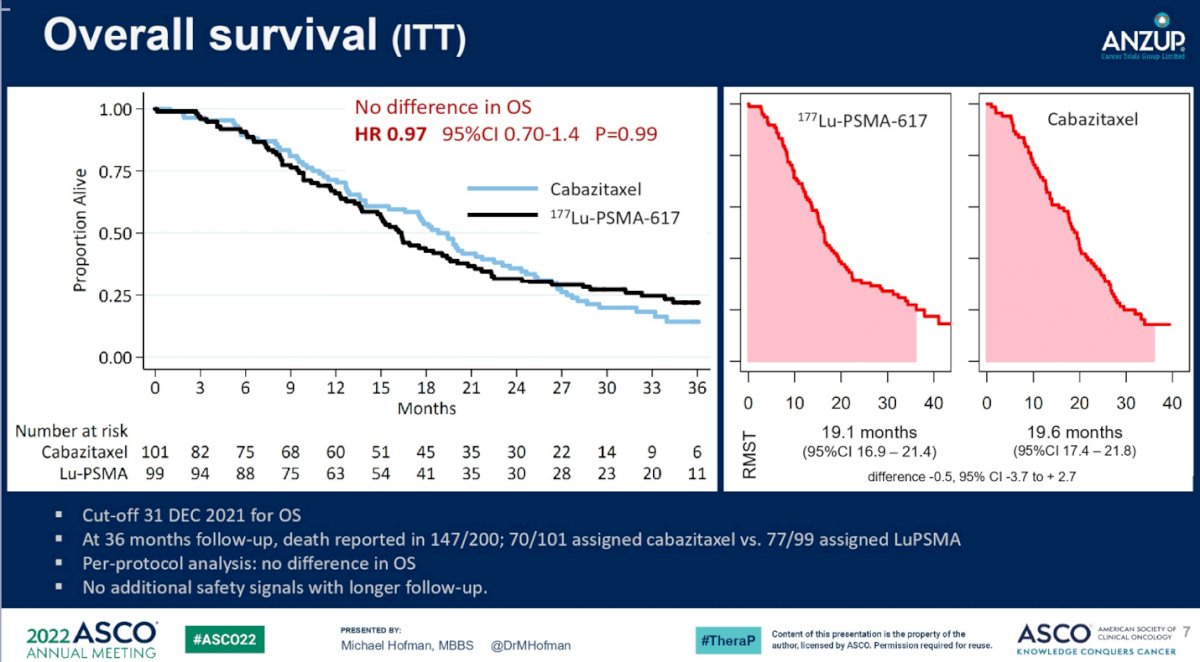
For the randomized patients, at a median follow up 36 months, death was reported in 70 of 101 patients assigned to receive cabazitaxel, 77 of 99 assigned to 177Lu-617, and 55 of 61 imaging-disqualified patients. In the cabazitaxel group, the two most common subsequent treatments were cabazitaxel (21%) and 177Lu-617 (20%). In the 177Lu-617 group, the most common subsequent treatment was cabazitaxel (32%)(shown as “Post Protocol System Treatment” on the figure below). Overall survival was found to not be different between the two groups (RMST to 36 months: 19.1 v 19.6 months, 95% CI -3.7 to 2.7). In this longer term follow up, no additional data safety concerns were raised.
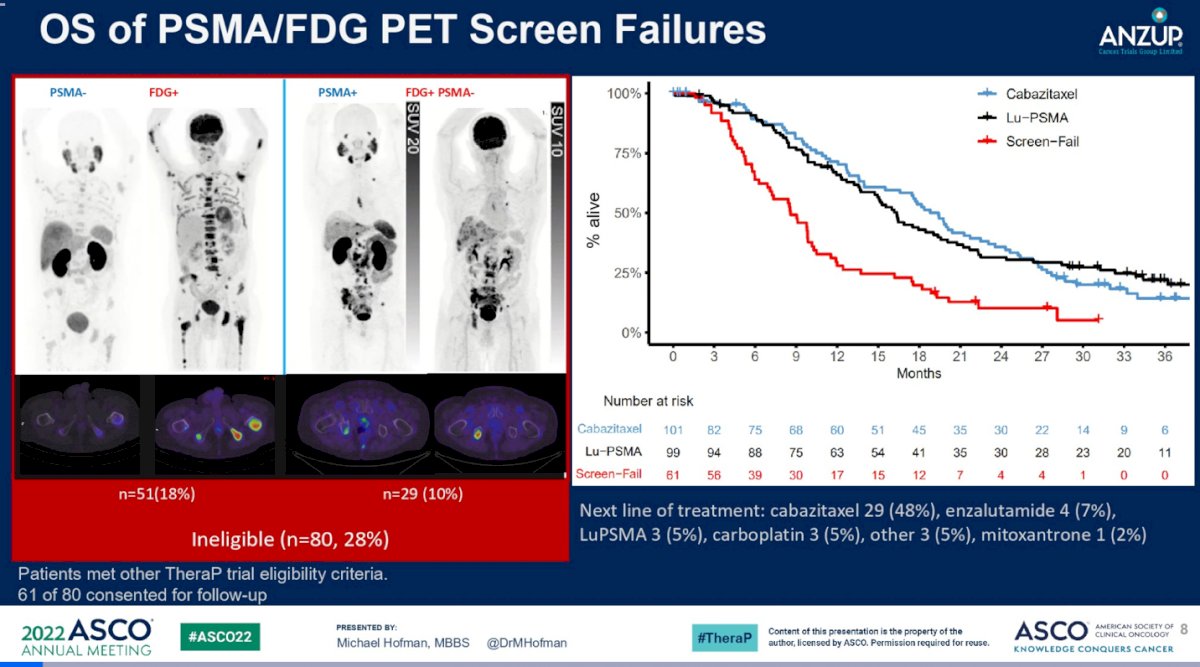
Among those 61 patients excluded based on their imaging, RMST over the same interval was 11.0 months (95% CI 9.0 to 13.1), noted be much shorter than the patients included in the study, irrespective of whether those patients received 177Lu-617 or cabazitaxel first. The inclusion of outcome data for those patients deemed by imaging criteria alone to be ineligible for the study offers a somewhat “real world” perspective on the deployment of PSMA targeted radiotherapy. These patients, among whom some did receive 177Lu-617, experienced shorter OS, perhaps reflecting aberrant biology of either PSMA-low or PSMA-heterogenous disease. Put another way, perhaps those patients with highest (and possibly uniform) PSMA expression should be prioritized for anti-PSMA targeted radiotherapy over chemotherapy. Whether PSMA-low could be overcome with a more potent radionuclide (e.g. alpha emitters such as 225Actinium) is an important question. In discussion, it was proposed that these patients also may benefit from intensification of chemotherapy with a doublet (e.g. carboplatin and cabazitaxel).
Although TheraP was never powered for overall survival, the OS signal is confounded by subsequent therapies which were used in similar proportions (Lu->Cabazi; Cabazi->Lu). He also spent some time contrasting to the phase III VISION study, including comparison to an active comparator (chemotherapy) and a higher PSMA PET cut off. Dr. Hofman concluded that treatment with 177Lu-PSMA-617 is a valid option for treatment of progressive mCRPC in patients who have already received docetaxel, as compared to cabazitaxel given response rates, improved PROs, and lower adverse event rates, with similar impact on overall survival. Much like other existing standard of care therapies for mCRPC, patient selection will depend on shared decision making with integration of imaging and circulating markers, patient prefences, and presumed disease biology (e.g. genomic study).
Presented by: Michael S. Hofman, MBBS (Hons), FRACP, FAANMS, Professor of Molecular Imaging, The University of Melbourne, and Nuclear Medicine Physician in the Centre for Cancer Imaging at the Peter MacCallum Cancer Centre in Melbourne, Australia
Written by: Jones Nauseef, MD, PhD, Assistant Professor of Medicine within the Division of Hematology and Medical Oncology, Sandra and Edward Meyer Cancer Center, and Englander Institute for Precision Medicine Weill Cornell Medicine and Assistant Attending physician at NewYork-Presbyterian Hospital. @DrJonesNauseef on Twitter during the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, Fri, June 3 – Mon, June 7, 2022.

