The 2022 ASCO annual meeting featured an oral abstract session on prostate cancer, including a presentation by Dr. Ian Davis discussing updated overall survival outcomes in ENZAMET (ANZUP 1304), an international, cooperative group trial of enzalutamide in mHSPC. Dr. Davis started by highlighting several points regarding the current mHSPC clinical knowledge. First, prognostic variables associated with better outcomes with testosterone suppression alone include (i) low volume being better than high volume disease, and (ii) metachronous metastatic presentation is better than synchronous. Second, the OS benefit of combination treatment by prognostic groups:
- Docetaxel + testosterone suppression > testosterone suppression alone: for synchronous and metachronous high volume patients
- Strong ADT (testosterone suppression + abiraterone/enzalutamide/apalutamide) > testosterone suppression alone: for all prognostic groups
- Radiation to the primary + testosterone suppression > testosterone suppression alone: for synchronous low volume disease
- Abiraterone or darolutamide + docetaxel + testosterone suppression > docetaxel + testosterone suppression: for when docetaxel is thought to be appropriate
The first planned interim analysis of ENZAMET, with 243 deaths after a median follow-up of 34 months, revealed a clinically meaningful overall survival (OS) benefit in mHSPC with the addition of enzalutamide to standard of care (HR 0.67, 95% CI 0.52 to 0.86, p=0.002 [1]). At ASCO 2022, Dr. Davis presented updated OS findings from the prespecified analysis triggered to occur after 470 deaths (cut-off date of January 19, 2022).
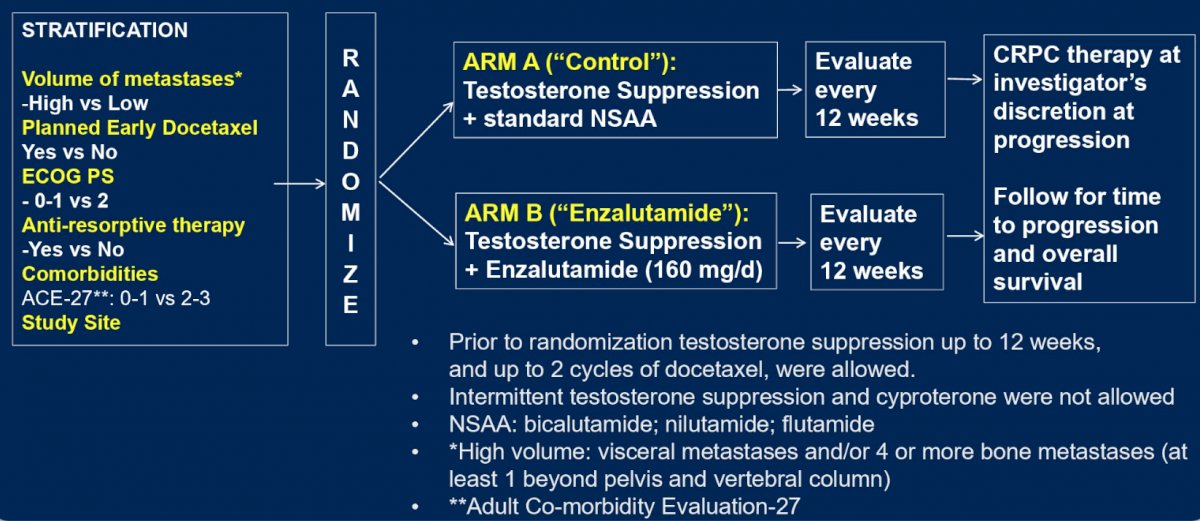
ENZAMET randomly assigned participants with mHSPC to treatment with testosterone suppression plus either a conventional non-steroidal anti-androgen or enzalutamide. Stratification factors included age, volume of disease (high vs low according to the CHAARTED definition), and planned use of concurrent docetaxel assigned by the treating physician (docetaxel yes vs no):
Longer term data included a median follow-up of 68 months, with several points to highlight from the prespecified in the statistical analysis plan: (i) at 470 deaths, no additional interim analysis will be performed, and (ii) identify synchronous versus metachronous M1: “M0 at primary diagnosis (Y/N)”. As an exploratory subgroup analysis, the question was asked: is the effect of enzalutamide modified by prognostic grouping and docetaxel use? Two binary factors were included for prognosis, including M1 at initial diagnosis (synchronous, de novo) or not (ie. not M0 or Mx), and high volume mHSPC at study entry, or not.
This trial randomized 1,125 patients with a median age of 69 years, including 503 in the docetaxel stratum, and 602 with high volume metastatic disease. OS results in the combined cohort are based on 476 deaths, with a median OS in the enzalutamide of not reached (95% CI not reached – not reached) compared to the control group of 73.2 months (95% CI 64.7 – not reached). The 5-year overall survival in the enzalutamide arm was 67% compared to 57% in the control arm:
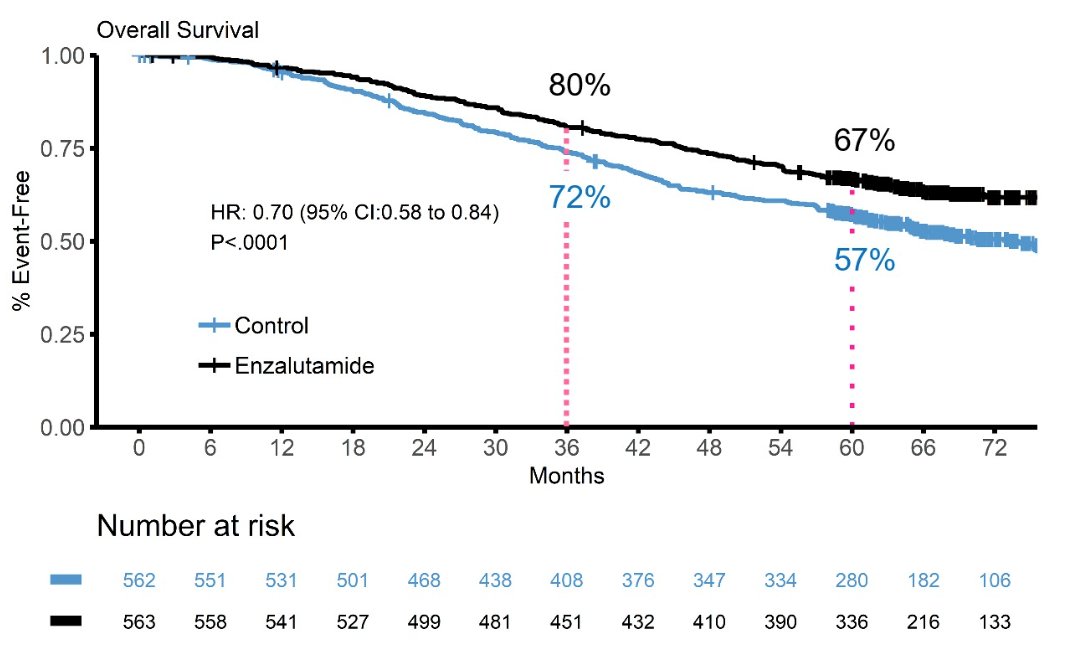
Participants continued on enzalutamide for longer than compared to the control group: median 57.8 months vs 22.6 months. Additionally, there was a substantial crossover in the control arm to enzalutamide or abiraterone for mCRCP, with 76% of those in the control arm receiving enzalutamide or abiraterone after progression, compared to 26% in the enzalutamide arm. Dr. Davis also highlighted several other key considerations:
- ENZAMET was representative, including all combinations of patient subgroups (synchronous, metachronous, high volume, low volume, use of docetaxel, etc), which were treated contemporaneously in the same trial
- Docetaxel use was at the investigator’s discretion, based on the assessment of “chemofitness”, with 45% of patients planned for concurrent docetaxel up to 6 cycles (median of 6). Before randomization, 108 received 1 cycle, and 62 received 2 cycles of docetaxel
- The trial design allows exploratory description of subgroup outcomes, but without formal comparisons due to confounding
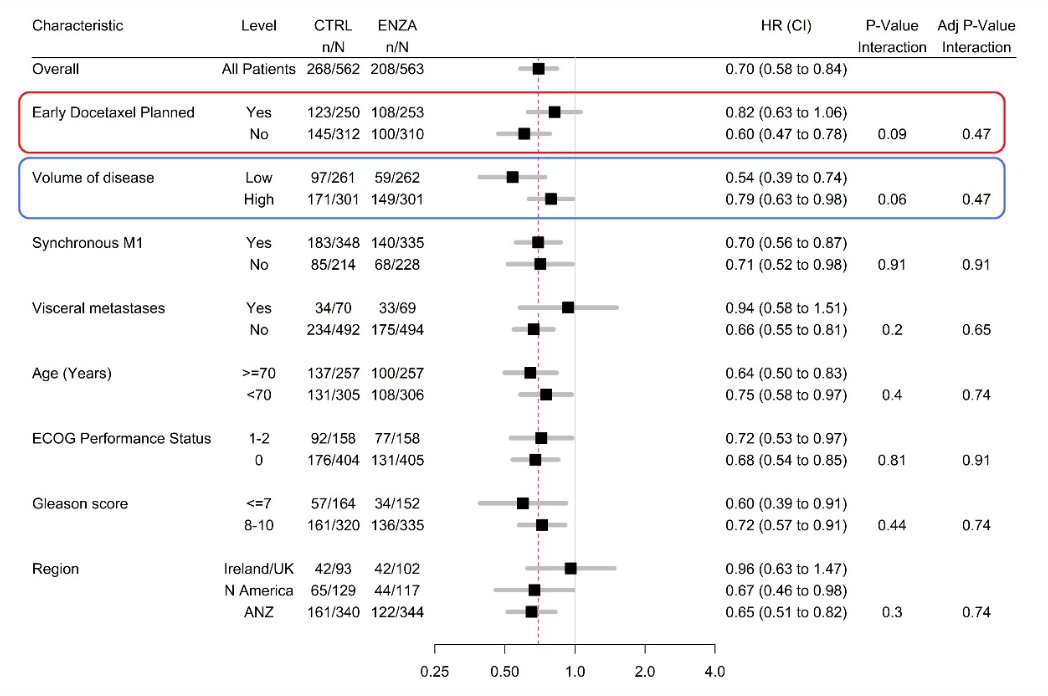
Overall survival by prespecified subgroup analysis are highlighted, with particular attention given to planned early docetaxel and volume of disease:
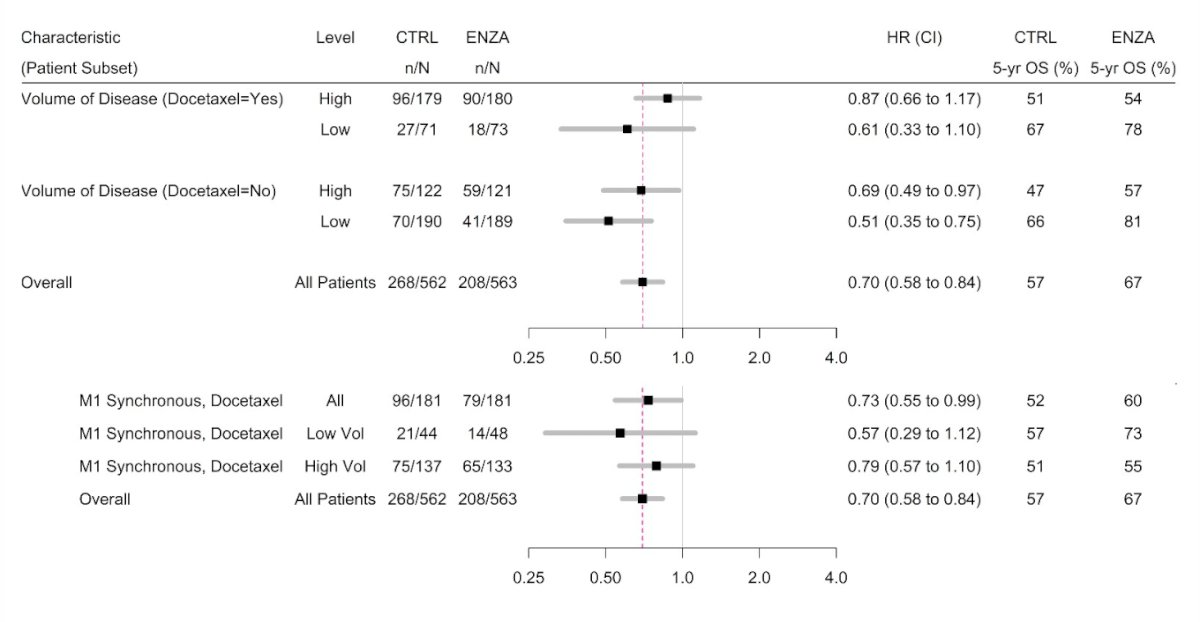
Overall survival based on volume, M1 timing and docetaxel also favored the enzalutamide arm:
The following Kaplan-Meier curves highlight the outcomes for PFS progression free survival:

Dr. Davis notes that the strengths of this study are (i) an active control arm with outcomes comparable to contemporary trials, (ii) concurrent use of docetaxel allowed as standard of care, (iii) there was a mix of synchronous and metachronous patients, as well as high and low volume patients, and (iv) there was a “hard” endpoint of overall survival. However, there are several limitations, including (i) docetaxel use was not randomized, (ii) the study was not powered for exploratory subset analysis, and (iii) we cannot confuse treatment effects with different prognostic groups. Dr. Davis’ clinical impressions for this updated ENZAMET analysis are as follows:
- There were no major differences found in enzalutamide efficacy across subgroups
- This confirms the benefit of enzalutamide in mHSPC, especially in low volume disease
- Exploratory analyses suggest additional benefit of enzalutamide when added to testosterone suppression + docetaxel
Dr. Davis concluded his presentation discussing updated overall survival outcomes in ENZAMET with the following take-home messages:
- Enzalutamide added to testosterone suppression for mHSPC, and compared with an active comparator of non-steroidal anti-androgen +/- docetaxel, provided clinically meaningful improvements in OS for the combined overall cohort, which persisted with an additional 3 years of follow-up
- The study is ongoing
- The benefits were more pronounced in patients with low volume mHSPC in those for whom docetaxel was not deemed necessary, and benefit was still apparent with synchronous high volume mHSPC where docetaxel was deemed necessary (despite a median overall survival of >60 months with testosterone suppression + docetaxel + non-steroidal anti-androgen)
- There are several hypotheses from the exploratory subgroup analyses:
- The greatest benefit of triplet therapy may be in those with the poorest prognosis disease (synchronous, high volume), and able to receive docetaxel
- For other subgroups, testosterone suppression + enzalutamide provides substantial increases in overall survival that are not augmented by concurrent docetaxel
Presented by: Ian Davis MB, BS, PhD, FRACP, FAChPM, medical oncologist, Professor of Medicine and Head of the Eastern Health Clinical School, Monash University, Box Hill, VIC, Australia
Co-Authors: Andrew James Martin, Robert Richard Zielinski, Alastair Thomson, Thean Hsiang Tan, Shahneen Sandhu, M. Neil Reaume, David William Pook, Francis Parnis, Scott A. North, Gavin M. Marx, John McCaffrey, Raymond S. McDermott, Nicola Jane Lawrence, Lisa Horvath, Mark Frydenberg, Simon Chowdhury, Kim N. Chi, Martin R. Stockler, Christopher Sweeney
Affiliations: NHMRC Clinical Trials Center, University of Sydney, Sydney, Australia, Orange & Dubbo Base Hospital, Orange, Australia, Royal Cornwall Hospital, Truro, United Kingdom, Royal Adelaide Hospital, Adelaide, Australia, Peter MacCallum Cancer Centre and Sir Peter MacCallum Department of Oncology, University of Melbourne, Melbourne, VIC, Australia, Ottawa Hospital Cancer Centre, Ottawa, ON, Canada, Monash Health, Melbourne, VIC, Australia, Ashford Cancer Center, Adelaide, Australia, Division of Medical Oncology, Cross Cancer Institute, University of Alberta, Edmonton, AB, Canada, Sydney Adventist Hospital, University of Sydney, Sydney, Australia, Mater Misericordiae University Hospital, Dublin, Ireland, St. Vincent's University Hospital, Dublin, Ireland, Auckland City Hospital, Auckland, New Zealand, Sydney Cancer Centre, Sydney, NSW, Australia, Monash University Faculty of Medicine, Clayton, Australia, Guy’s, King's, and St. Thomas' Hospitals, London, United Kingdom, BC Cancer Agency, University of British Columbia, Vancouver, BC, Canada, NHMRC Clinical Trials Centre, University of Sydney, Sydney, NSW, Australia, Lank Center for Genitourinary Oncology, Dana-Farber Cancer Institute, Boston, MA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, Fri, June 3 – Mon, June 7, 2022.
References:
Related Content: Enzalutamide with Standard First-Line Therapy in Metastatic Prostate Cancer (ENZAMET) - Tanya Dorff

