(UroToday.com) At the 2022 American Society of Clinical Oncology Annual Meeting held in Chicago and virtually, the oral abstract session focused on Prostate, Testicular, and Penile cancers on Sunday morning included a presentation from Dr. Andrew J. Armstrong standing in for Phillip Kuo discussing the prognostic value of baseline [68Ga]Ga-PSMA-11 PET imaging in men undergoing theranostic treatment with 177Lu-PSMA-617 for heavily pre-treated metastatic castration-resistant prostate cancer (mCRPC) in the VISION trial.
Theranostic treatment using 177Lu-PSMA-617has rapidly gained interest in both research and clinical practice spheres. The principle of theranostic approaches rely on targeted delivery of treatment directly to cancer cells: in the case of 177Lu-PSMA-617, the beta-emitter lutetium is targeted to prostate cancer cells using antibodies to the transmembrane protein PSMA (prostate specific membrane antigen).
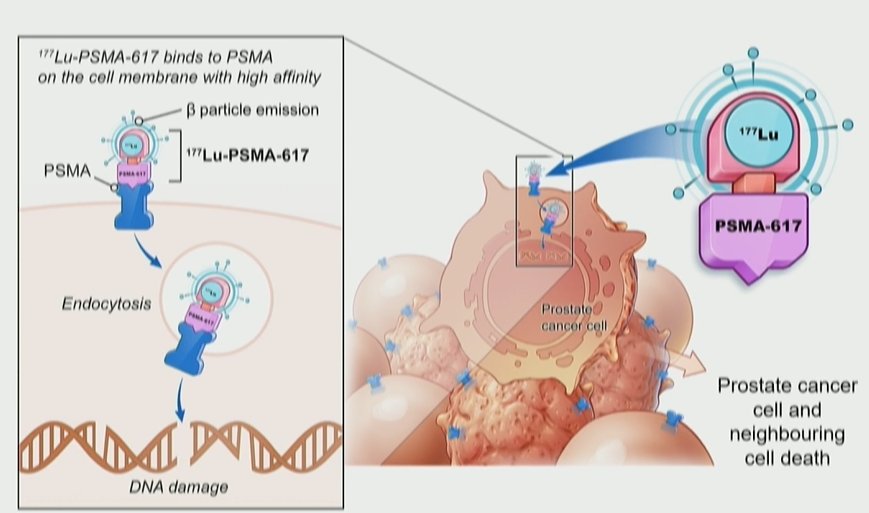
The VISION trial (NCT03511664) provided the first phase III randomized controlled evidence to support the use of LuPSMA and led to its recent FDA approval. VISION used gallium (68Ga) gozetotide (68Ga-PSMA-11) PET/CT imaging at enrollment to determine eligibility for lutetium (177Lu) vipivotide tetraxetan (177Lu-PSMA-617).
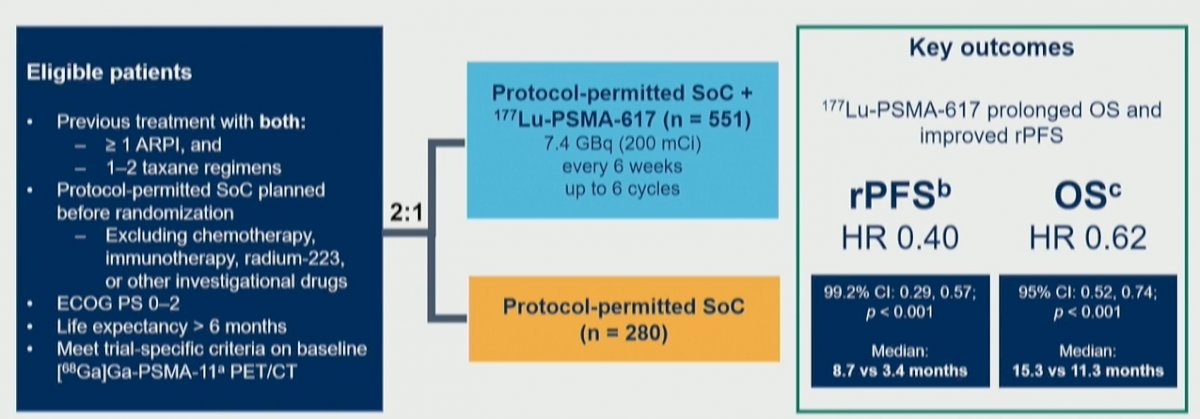
Given that 177Lu-PSMA-617 targets PSMA, Dr. Kuo and colleagues assessed the association between quantitative PSMA imaging parameters and treatment outcomes.
To briefly summarize, the VISION trial enrolled adult patients who had previously received treatment with at least 1 androgen receptor pathway inhibitor (ARPI) and 1–2 taxane regimens for mCRPC. These men were randomized 2:1 to 177Lu-PSMA-617 (7.4 GBq Q6W, up to 6 cycles) + SoC or SoC alone. Protocol-permitted SoC excluded cytotoxic chemotherapy, systemic radioisotopes, immunotherapy, or other investigational drugs.
In terms of imaging inclusion and exclusion criteria, men had to have at least one 1 PSMA-positive (+) and no PSMA-negative lesions.

In this sub-study of the VISION trial, the authors assessed the association between imaging data from pre-enrollment 68Ga-PSMA-11 PET/CT scans of patients in the 177Lu-PSMA-617 group and clinical outcomes.

Among 551 potentially eligible patients, 548 had imaging data that met quality requirements. PSMA expression was quantified by 5 PET parameters: PSMA+ lesions by region, mean standardized uptake value (SUVmean), maximum SUV (SUVmax), PSMA+ tumor volume, and tumor load (PSMA+ tumor volume × SUVmean). Parameters were extracted from the whole body and 4 regions.
Before discussing results, Dr. Armstrong provided definition of these terms. He first noted that the SUV represents the ratio of active per unit volume of a region of interest to the active per unit whole-body volume. SUVmean refers to the mean number of counts from all voxels within the whole body, with all PSMA positive lesions pooled together. SUVmax represents the level of uptake in the voxel with the higher number of counts in the whole body. Tumor volume denote the total volume of PSMA positive lesions as a sum of the total volume of voxels with positive uptake. Tumor load is a relatively novel concept, combining both volume and intensity in the whole body. Finally, the authors assessed the binary presence of PSMA-positive lesions within discrete segmented anatomic regions.
The association between PET parameters and clinical outcomes including radiographic progression-free survival (rPFS; primary objective), overall survival (OS), objective response rate (ORR), and prostate–specific antigen 50 (PSA50) response was assessed.
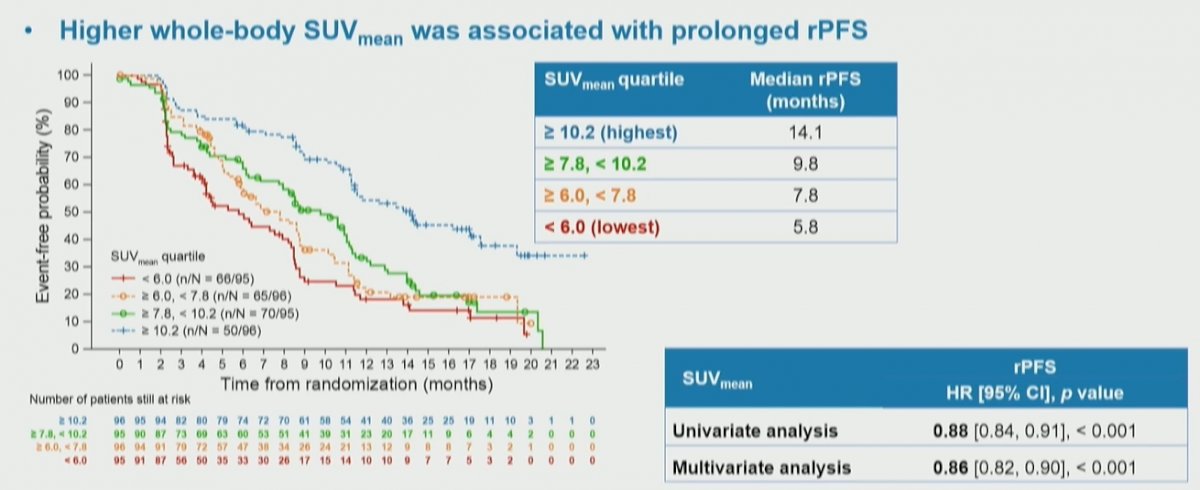
Among the 548 evaluable patients, most (92.7%) had PSMA uptake in bone and 13.1% had uptake in the liver. In both the whole-body and regional analyses, statistically significant associations of PSMA PET parameters to clinical outcomes were observed. However, these were not consistent. He noted that SUVmean provided a consistent statistically significant association with improved response across all endpoints assessed.

In more detail, higher whole-body SUVmean was associated with improved clinical outcomes: those patients in the highest quartile (SUVmean: rPFS, ≥ 10.2; OS, ≥ 9.9) had a median rPFS and OS of 14.1 and 21.4 months, vs 5.8 and 14.5 months for those in the lowest quartile (< 6.0; < 5.7), respectively.
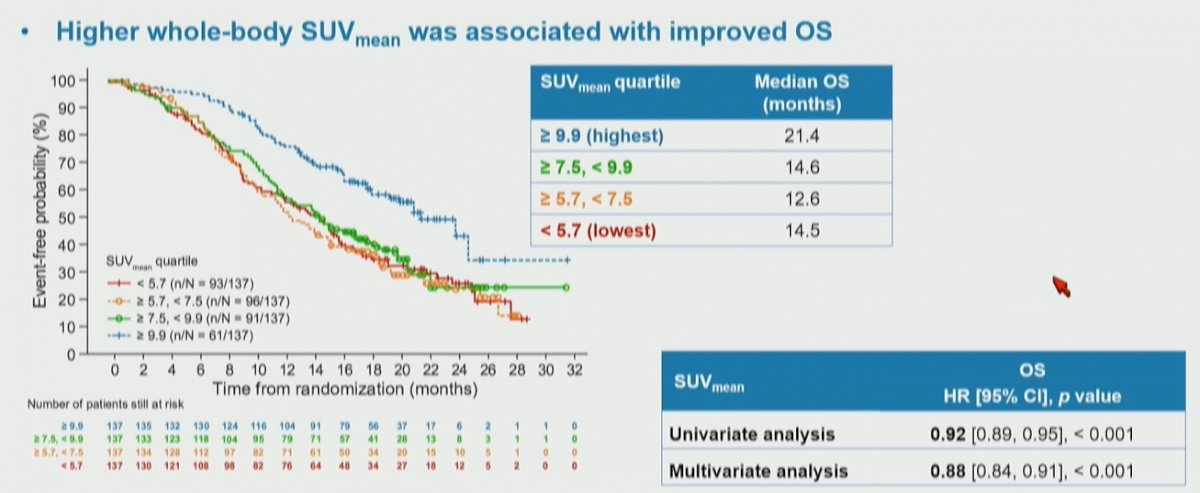
The authors then assessed the association of these quartiles of whole body SUVmean with overall survival, again finding a significant stratification between the groups. Notably, among these patients who received 177Lu-PSMA-617, those with the highest SUVmean levels had the longest overall survival.
Further, patients with liver metastases have a particularly poor prognosis. Thus, not surprisingly, patients with the absence of PSMA+ lesions in bone and liver particularly had improved overall prognosis and those with lymph node only metastases had the best prognosis.

Thus, the authors conclude that a higher SUVmean is strongly associated with improved outcomes for men with mCRPC undergoing theranostic treatment with 177Lu-PSMA-617. Additionally, the absence of PSMA positive disease in the liver and bone was associated with improved outcomes. These data support the use of 68Ga-PSMA-11 to identify men with mCRPC who most benefit from PSMA-targeted theranostic treatment. Further work is ongoing to better define those subgroups of patients who most benefit from this treatment approach.
Presented by: Andrew J. Armstrong, MD, MSc, Medical Oncologist, Duke Health

