(UroToday.com) The 2022 ASCO annual meeting featured an oral abstract session on kidney and bladder cancer, including a presentation by Dr. Monty Pal discussing results of cohorts 3, 4, and 5 in the COSMIC-021 study assessing cabozantinib + atezolizumab in urothelial carcinoma. Cabozantinib, a multi-targeted receptor tyrosine kinase inhibitor (TKI), promotes an immune-permissive environment that may enhance response to immune checkpoint inhibitors. Atezolizumab is approved as monotherapy for cisplatin ineligible and PD-L1 positive or platinum-containing chemotherapy-ineligible locally advanced or metastatic urothelial carcinoma. As such, COSMIC-021, a multicenter phase 1b study, is evaluating cabozantinib + atezolizumab (anti‒PD-L1 therapy) in various solid tumors (NCT03170960). Cabozantinib + atezolizumab demonstrated encouraging clinical activity in cohort 2 of COSMIC-021 in patients with urothelial carcinoma previously treated with platinum-containing chemotherapy (presented by Pal S et al., ASCO 2020). Outcomes of cabozantinib + atezolizumab from three other urothelial carcinoma cohorts (C3, C4, C5) were presented at ASCO 2022 by Dr. Pal.
This study included patients with inoperable locally advanced/metastatic urothelial carcinoma with transitional cell histology and ECOG PS 0‒1. Patients enrolled in C3 and C4 had no prior therapy and were cisplatin-based chemotherapy ineligible (C3) or eligible (C4). C5 enrolled patients with one prior immune checkpoint inhibitor and no prior VEGFR-TKI therapy. Patients received cabozantinib 40 mg PO QD and atezolizumab 1200 mg IV every 3 weeks. CT/MRI scans were performed every 6 weeks for first year and every 12 weeks thereafter. The primary endpoint of this study was objective response rate per RECIST v1.1 by investigator. Other endpoints included safety, duration of response (DOR), PFS, and OS. The study schema for COSMIC-021 is as follows:

There were 30 patients each enrolled in C3 and C4, and 31 in C5. Baseline characteristics for C3, C4, and C5 were, respectively: median age, 74 years of age, 66 years of age, and 68 years of age; male, 67%, 73%, 55%; ECOG PS 1, 63%, 57%, 74%; lung/liver metastasis; 33%/17%, 40%/20%, 58%/23%; ≥3 tumor sites, 30%, 43%, 45%; bladder as primary site, 67%, 70%, 71%:
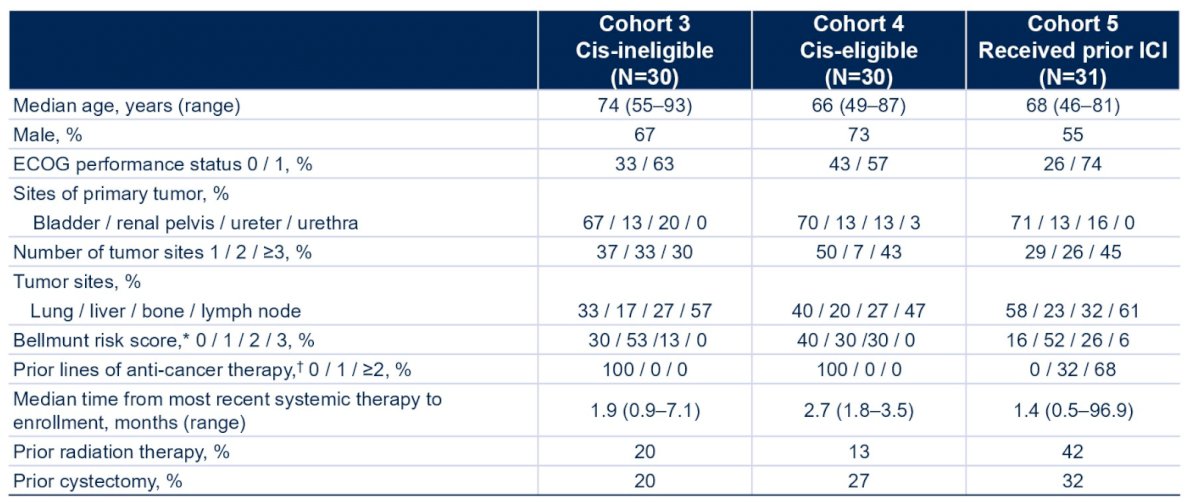
As of Nov 30, 2021, the median follow-up for C3, C4, and C5 was 27.9, 19.1, and 32.9 months, respectively, with 1, 6, and 1 patients in each cohort remaining on treatment. Cabozantinib + atezolizumab demonstrated clinical benefit across all cohorts: the objective response rate for C3 was 20% (95% CI 8-39), for C4 was 30% (95% CI 15-49%), and for C5 was 10% (95% CI 2-26%). The disease control rate for C3 was 80% (95% CI 61-92), for C4 was 63% (95% CI 44-80%), and for C5 was 61% (95% CI 42-78%):

The median duration of response for C3 was 7.1 months, for C4 was not reached, and for C5 was 4.1 months. The change in sum of target lesions over time is as follows:
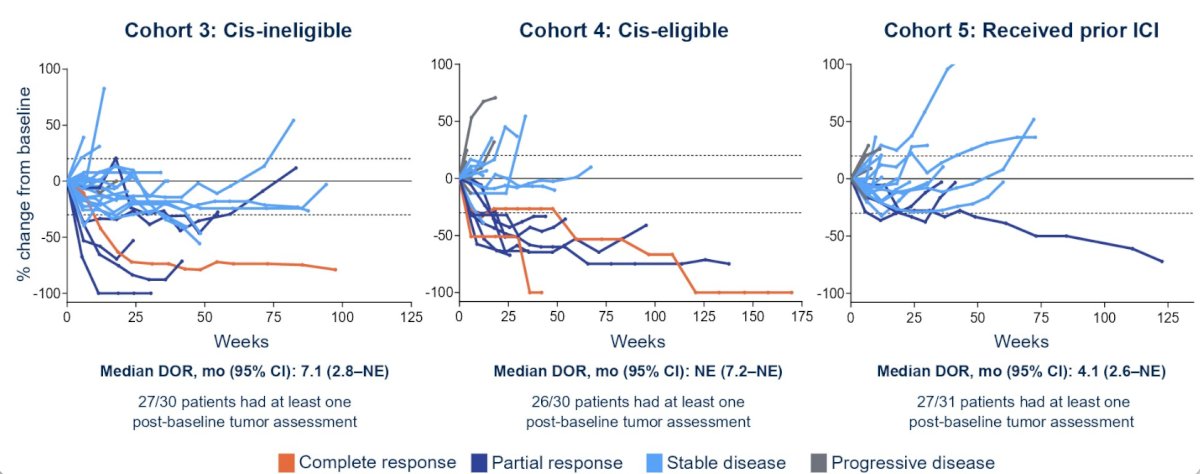
With regards to PFS, median PFS for C3 was 5.6 months (95% CI 3.1-11.1), for C4 was 7.8 months (95% CI 1.6-13.8), and for C5 was 3.0 months (95% CI 1.8-5.5). For OS, the median OS was 14.3 months for C3 (95% CI 8.6 – not reached), for C4 was 13.5 months (95% CI 7.8-23.2), and for C5 was 8.2 months (95% CI 5.5-9.8). The most common treatment-related adverse events of any grade across C3, C4, and C5, respectively, were diarrhea (43%, 33%, 35%), nausea (27%, 17%, 26%), fatigue (27%, 27%, 48%), and decreased appetite (33%, 27%, 39%); grade 3/4 treatment-related adverse events occurred in 63%, 43%, and 45%, and there was no grade 5 treatment-related adverse events.
Dr. Pal concluded his presentation by discussing the results of cohorts 3, 4, and 5 in the COSMIC-021 study assessing cabozantinib + atezolizumab in urothelial carcinoma with the following take-home messages:
- Cabozantinib + atezolizumab demonstrated encouraging clinical activity with manageable toxicity for patients:
Presented by: Sumanta K. Pal, MD, City of Hope Comprehensive Cancer Center, Duarte, CA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting, Chicago, IL, Fri, June 3 – Mon, June 7, 2022.

