Regardless, a post-hoc analysis of pooled Phase 3 data of sipuleucel-T (n = 737) suggested substantial overall survival (OS) benefit for AA men receiving sipuleucel-T (n = 33) vs placebo (n = 10).1 Compared with pooled placebo patients (n = 249), the number needed to treat for overall survival (OS) benefit at 3 years was 3 for AAs and 8 for all sipuleucel-T-treated patients (n = 488).2 If this is true, this would be an important treatment option for asymptomatic or minimally symptomatic mCRPC AA men. Though, it should be noted that sipuleucel-T is expensive and not used clinically as much as other treatment options in this disease space.
In this study, Dr. Oliver Sartor and colleagues analyzed PROCEED, a large real-world registry, in which all patients received sipuleucel-T. In PROCEED, 1902 mCRPC patients received ≥1 sipuleucel-T infusion (typically men receive 3 doses, every two weeks). The main endpoint of this analysis was OS — OS of all AA (n = 221) and CAU (n = 1649) men were compared. However, baseline prostate-specific antigen (PSA), the most important prognostic variable for OS after sipuleucel-T (Schellhammer 2013), substantially differed by race. Thus, OS for a PSA-matched cohort (n = 219 AA; n = 438 CAU) was compared and univariable/multivariable analyses were performed. PSA was matched to within 10%. Post-sipuleucel-T use of OS-prolonging anticancer interventions was also assessed.
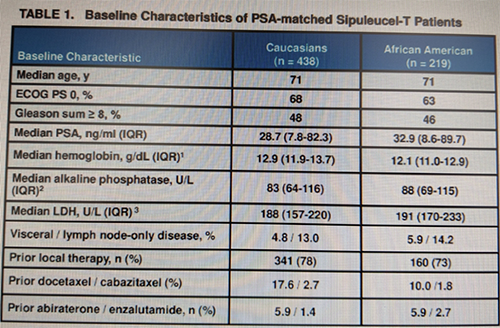
Full demographics of both groups is seen below:
Post-sipuleucel-T life-prolonging anti-cancer therapies were balanced between groups.
After a median follow-up of 46.6 months (~4 years), median OS was 35.2 (all sipuleucel-T-treated AAs) and 29.9 months (all sipuleucel-T-treated CAUs): HR 0.81, 95% CI 0.68–0.97; P = 0.03.
In the PSA-matched cohort, median OS was 35.3 and 25.8 mo, respectively (HR 0.70, 95% CI 0.57–0.86; P < 0.001). The KM curve is seen below:
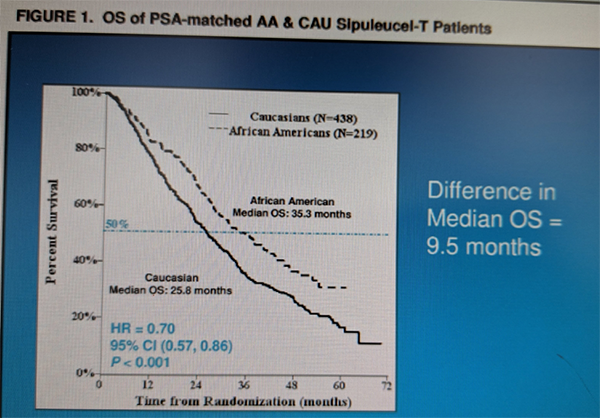
Interestingly, even without PSA matching, OS appeared to be higher in the AA cohort.
Sipuleucel-T-treated AAs with lower baseline PSA had markedly longer median OS vs sipuleucel-T-treated CAUs. Among those with ≤ median baseline PSA (29.48 ng/ml), median OS was 54.3 mo (AA) vs. 33.4 (CAUs); HR 0.52, 95% CI 0.37–0.72; p < 0.001. When broken down by quartiles, the data is as below:
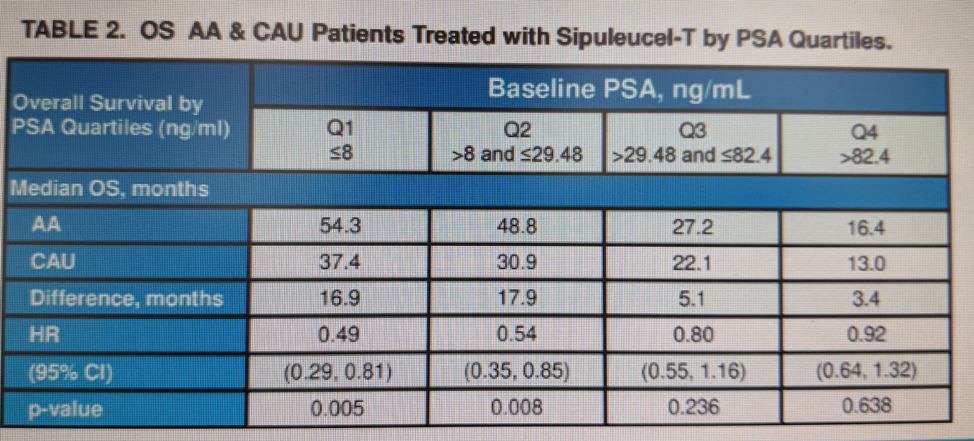
Those in the lowest 2 quartiles benefited, while those with higher baseline PSA’s did not benefit anymore than their Caucasian counterparts.
On MV analysis, along with other known prognostic factors, AA race was independently associated with prolonged OS on detailed multivariable analyses (HR 0.60, 95% CI 0.48–0.74; p < 0.001) and confirmed on sensitivity analyses. The full model is seen below:

Based on the strength of this real-world data, in addition to the prior phase III trial data, it would appear that Sipuleucel-T-treated AAs had significantly improved OS vs sipuleucel-T-treated CAUs. This analysis marks the largest known racial difference in OS in response to any therapy for mCRPC, a finding with implications for both prostate cancer pathophysiology and cancer immunotherapy.
Presented by: A. Oliver Sartor, MD, Professor of Medicine, Medical Director, Tulane Cancer Center, C. E., and Bernadine Laborde Professor of Cancer Research, Tulane University, New Orleans, Louisiana, United States
Co-Authors: Andrew J. Armstrong, Duke Cancer Institute Center for Prostate and Urologic Cancers, Durham, NC; Chiledum Ahaghotu, MD, Howard University Hospital, Washington, DC; David G. McLeod, MD, Walter Reed National Military Medical Center, Bethesda, MD; Matthew R. Cooperberg, MD, MPH, University of California, San Francisco, San Francisco, CA; David F. Penson, MD, MPH, MMHC, Vanderbilt University Medical Center, Nashville, TN; Philip W. Kantoff, MD, Department of Medicine, Memorial Sloan Kettering Cancer Center, New York, NY; Nicholas J. Vogelzang, MD, FASCO, FACP, Comprehensive Cancer Centers of Nevada, Las Vegas, NV; Arif Hussain, MD, University of Maryland Cancer Center, Baltimore, MD; Christopher Michael Pieczonka, MD, Associated Medical Professionals of New York, PLLC, Syracuse, NY; Neal D. Shore, MD, FACS, Carolina Urologic Research Center, Myrtle Beach, SC; David I. Quinn, MD, USC Norris Comprehensive Cancer Center, Los Angeles, CA; Eric Jay Small, MD, FASCO, UCSF Helen Diller Family Comprehensive Cancer Center, San Francisco, CA; Elisabeth I. Heath, MD, Barbara Ann Karmanos Cancer Institute, Wayne State University, Detroit, MI; Ronald F Tutrone, MD, FACI, Chesapeake Urology, Towson, MD; Paul F. Schellhammer, MD, Urol of Virginia, Virginia Beach, VA; Matthew Harmon, MD, Dendreon Pharmaceuticals LLC, Seattle, WA; Nancy N. Chang, MD, Cedars-Sinai Medical Center, Los Angeles, CA; Stephen J. Freedland, MD, University of Washington and Fred Hutchinson Cancer Research Center, Seattle, WA; Celestia S. Higano, MD, University of Washington and Fred Hutchinson Cancer Research Center, Seattle, WA.
Written by: Thenappan Chandrasekar, MD, Clinical Instructor, Thomas Jefferson University, @tchandra_uromd, @JEFFUrology, at the 2019 ASCO Annual Meeting #ASCO19, May 31- June 4, 2019, Chicago, IL USA
References:
- McLeod et al. AUA 2012.
- Moses et al. GU ASCO 2019.

