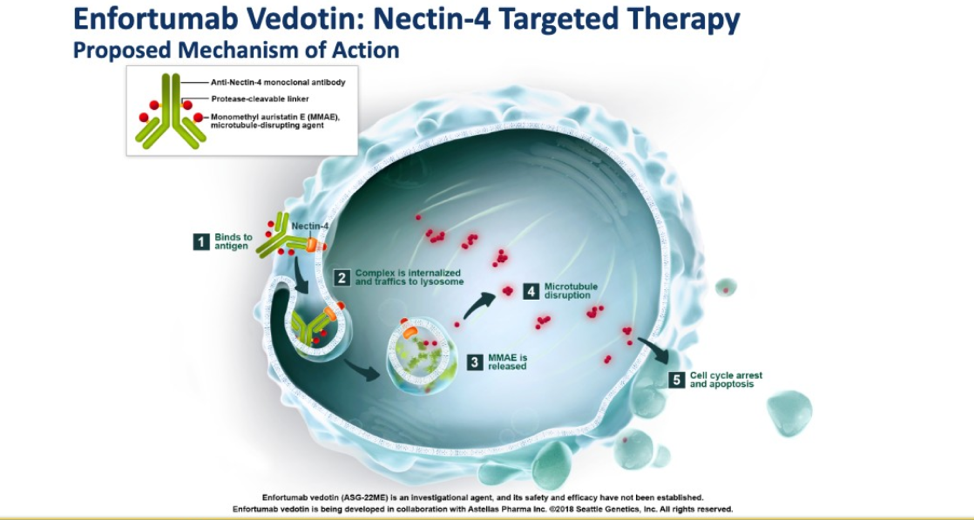
Chicago, IL (UroToday.com) After cisplatin-based chemotherapy and immune checkpoint inhibitors, there exist a paucity of effective therapies for patients with metastatic urothelial carcinoma (mUC). Enfortumab vedotin (EV) is an antibody-drug conjugate (ADC) which delivers a microtubule-disrupting agent to tumors expressing Nectin-4, a protein overexpressed in urothelial, breast, pancreatic, lung, and esophageal cancer.1
In the phase I EV-101 study of enfortumab vedotin, 112 patients received EV, the majority of whom received prior platinum chemotherapy (81%) and a checkpoint inhibitor (75%) and this patient population had an objective response rate of 33%. The median overall survival (OS) was reported to be 12.5 months.2 In this study, the authors describe the results of EV-201, EV monotherapy for locally advanced or metastatic urothelial cancer previously treated with platinum and immune checkpoint inhibitors.
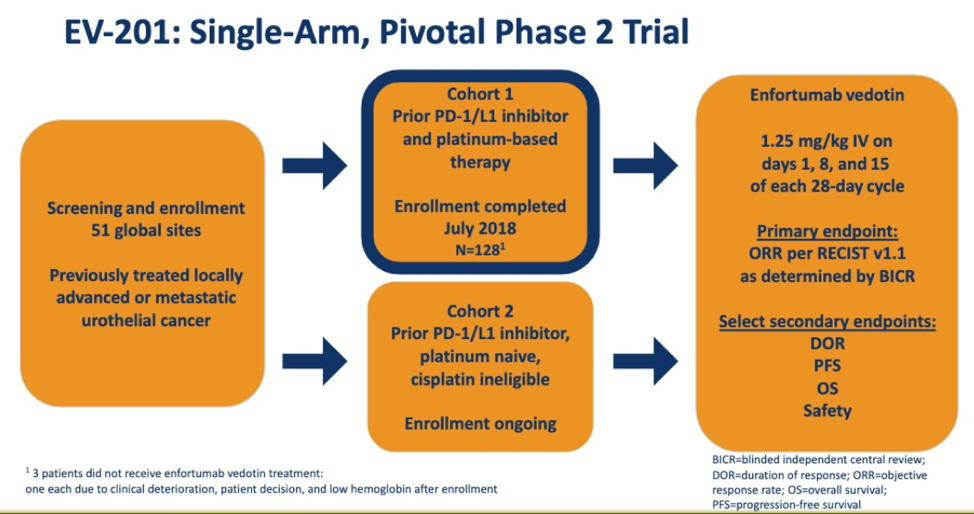
This abstract provides data on Cohort 1 of the study above. 125 patients were enrolled and the maximum time on treatment is 15.6 months and ongoing. Eligibility criteria are shown below.
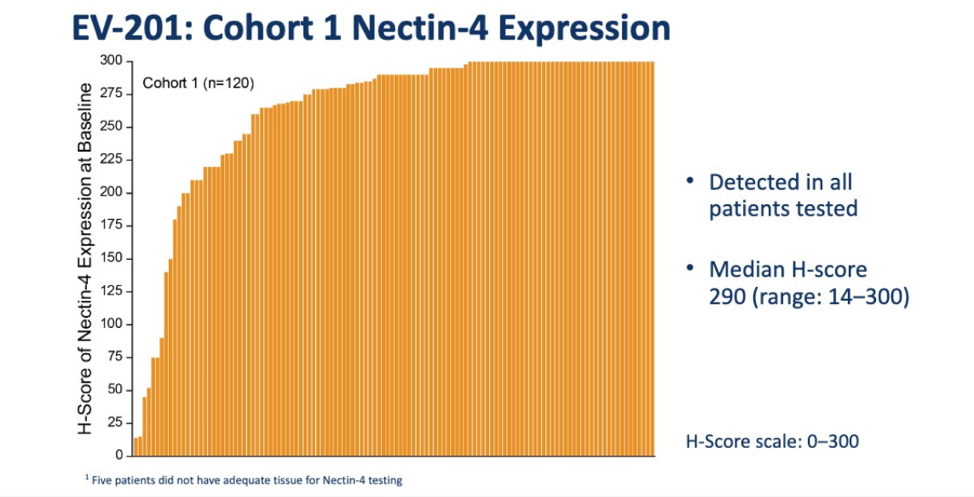
Of note, nectin expression was NOT mandated in the study but was measured for all patients and could be detected in ALL patients.
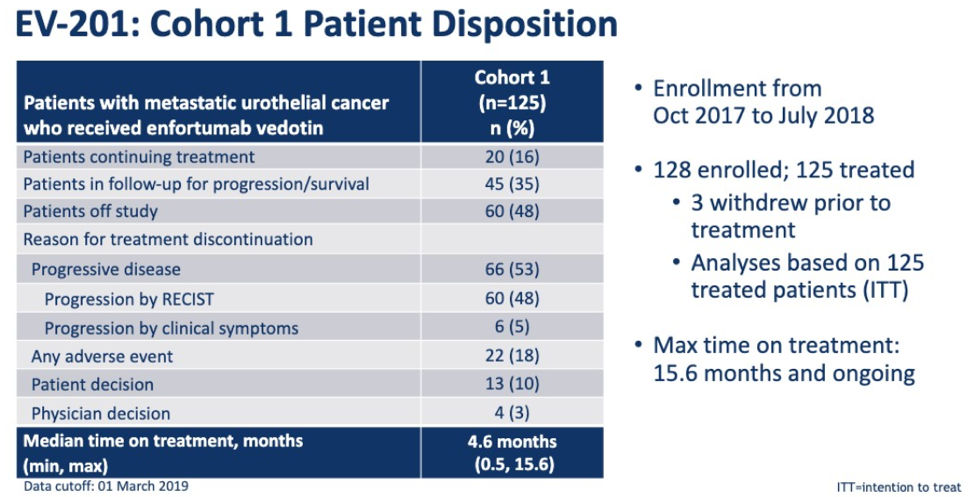
125 patients were enrolled and 16% of patients continue on treatment. The median time of treatment was 4.6 months and 53% of patients have discontinued due to the progression of disease.
Baseline characteristics are typical of this population of patients with mUC. Of note, 42% of patients had 2 or more Bellmunt adverse prognostic factors and 27% of patients were 75 or older. 35% of patients had upper tract disease.
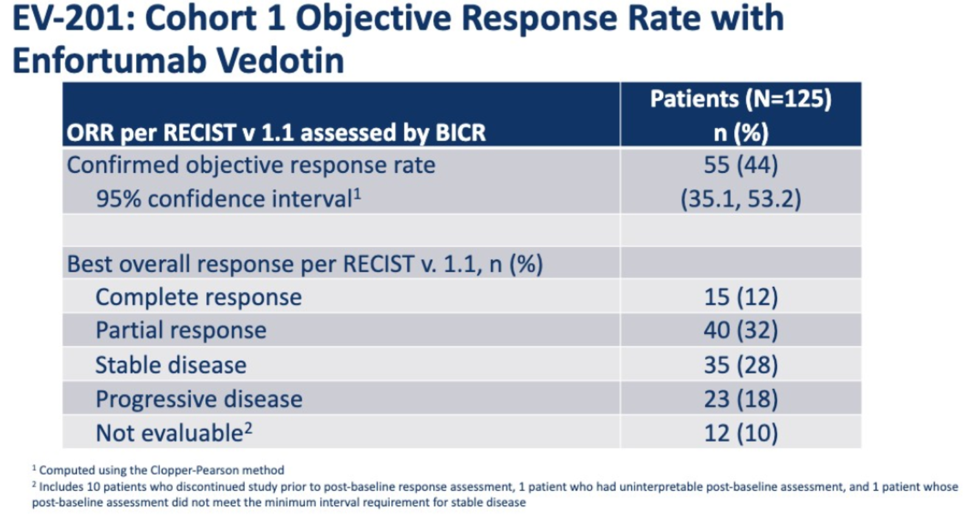
In terms of response, 44% of patients had a confirmed objective response, with a 12% complete response (CR) rate.
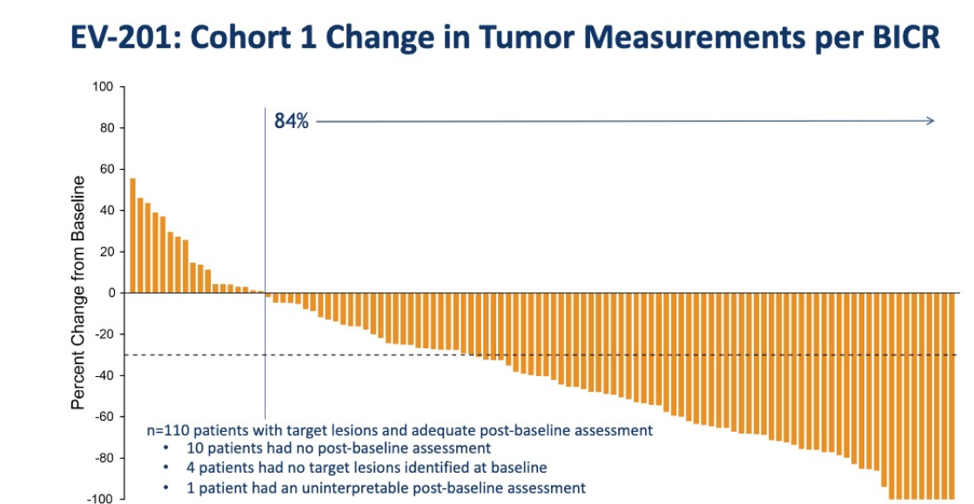
84% of all patients had tumor shrinkage.
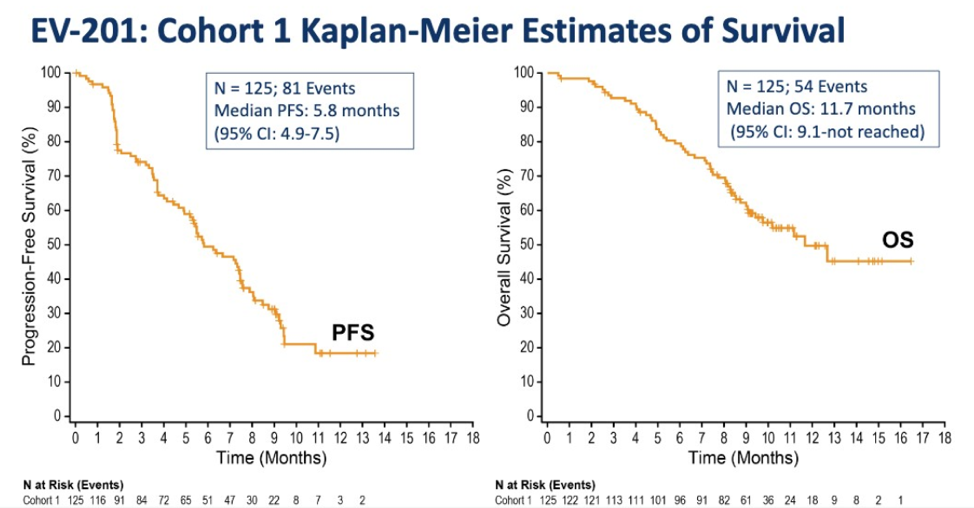
In terms of progression-free survival (PFS) and overall survival (OS), the median OS is 11.7 months in this phase 2 with a PFS of 5.8 months.
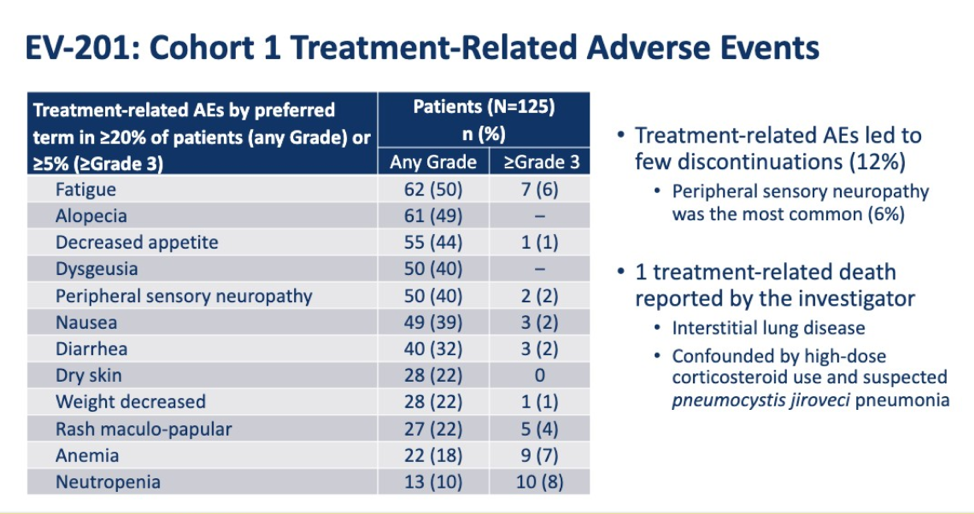
In terms of AEs, 12% of patients discontinued due AEs, with the most common reason being neuropathy. There was one
treatment-related death but this was in a patient with ILD on high dose steroids who may have had PCP pneumonia. The most common grade 3 AEs were fatigue, anemia, and neutropenia.
In this single-arm phase II study, enfortumab vedotin shows promising anti-tumor activity in an unselected population of patients with mUC who have had either prior PD-1/L1 or prior platinum therapy. This was a high-risk population, many with visceral metastases, and despite this enfortumab showed great efficacy with a 12% CR rate. We look forward to seeing the data in cohort-2, which will include cisplatin-ineligible patients as well as data from EV-301, the randomized phase 3 study comparing enfortumab to standard of care post-platinum.
Presented by: Daniel Peter Petrylak, MD, Yale Cancer Center, New Haven, CT
Written by: Jason Zhu, MD. Fellow, Division of Hematology and Oncology, Duke University, @TheRealJasonZhu at the 2019 ASCO Annual Meeting #ASCO19, May 31- June 4, 2019, Chicago, IL USA
References:
- Challita-Eid PM, Satpayev D, Yang P, et al. Enfortumab vedotin antibody–drug conjugate targeting nectin-4 is a highly potent therapeutic agent in multiple preclinical cancer models. Cancer research 2016;76:3003-13.
- Rosenberg JE, Sridhar SS, Zhang J, et al. Updated results from the enfortumab vedotin phase 1 (EV-101) study in patients with metastatic urothelial cancer (mUC). American Society of Clinical Oncology; 2018.