In the SWOG 8710 trial,1 the first randomized study to show a benefit for neoadjuvant chemotherapy in muscle-invasive bladder cancer patients, the rate of pathological stage T0 was 38% in the chemotherapy arm and 15% in the control arm.
The NCI-60 consists of 60 cell lines from nine common cancer types and provides a rich dataset for evaluating gene expression profiling of drug sensitivity. COXEN represents a “correlation of correlations,” allowing for the application of in vitro NCI-60 results to individual patients. The first step in this process is the identification of the gene expression associated with individual drug sensitivity in the NCI-60. These genes are then compared with the application set to identify concordant genes. By comparing the NCI-60 results with the gene expression of the target findings, a correlation coefficient (COXEN coefficient) is derived.
In the presented trial (study design in Figure 1) eligible patients were randomized to either GC or dose-dense MVAC and then underwent radical cystectomy with pelvic lymph node dissection. Eligible patients were T2-T4a N0M0 urothelial carcinoma patients. None of the patients received prior chemotherapy for urothelial carcinoma. The performance status was 0 or 1, the creatinine clearance was ≥60 ml/min, and patients could not have any hearing impairment. The primary objective was to characterize the relationship of dense dose MVAC- and GC- specific COXEN scores in terms of pathological stage T0 rate at cystectomy in patients treated with neoadjuvant chemotherapy. This was performed by assessing whether the COXEN score was prognostic of pathological T0 rate or ≤ pathological stage T1. The authors also assessed, in a preliminary fashion, whether the COXEN score is a predictive factor distinguishing between these two chemotherapy regimens. Secondary objectives included:
- Assessing the value of gene expression profiling in predicting overall survival (OS) in bladder cancer patients treated with neoadjuvant chemotherapy
- Assessing the difference in pathological stage T0 rate between the 21-day GC and 14-day dose-dense MVAC arms, regardless of gene expression.
- Assessing the safety and tolerability of both chemotherapy regimens
Figure 1 – S1314 study design:

Figure 2 – CONSORT diagram:
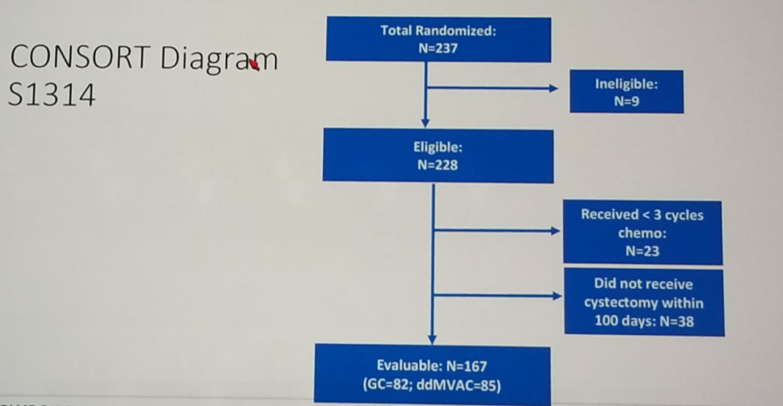
Table 1 – Baseline characteristics:

Table 2 – Descriptive data on pathologic response rate by treatment arm:

Table 3 – Results of the primary analysis:

The results demonstrated that COXEN was not predictive of response by treatment arm. As a phase two study, there was low statistical power to detect this. It was shown that GC was predictive of pathologic response to chemotherapy in a pooled analysis of patients from both arms.
Dr. Flaig continued and stated that the S1314 study is a platform to examine other biomarkers of interest beyond COXEN in muscle-invasive bladder cancer.
In conclusion, GC and dose-dense MVAC COXEN scores were not significantly predictive for response in their individual arms. Unfortunately, the number of patients accrued was below the required number. The COXEN GC score was shown to be a significant predictor for downstaging when combining subjects in the GC and dose-dense MVAC arms. Lastly, there was no evidence of an interaction between COXEN score and treatment regimen in predicting response.
The authors plan to perform additional secondary analyses, including Cisplatin only COXEN predictor, increase the number of analyzed patients, and to report the results of other secondary objectives.
Presented by: Thomas W. Flaig, MD, Division of Medical Oncology, School of Medicine, University of Colorado Denver, Aurora, CO
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, @GoldbergHanan at the 2019 ASCO Annual Meeting #ASCO19, May 31- June 4, 2019, Chicago, IL USA
References:


