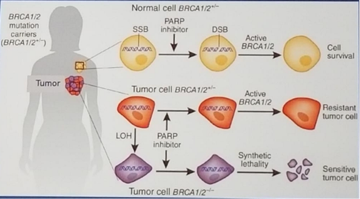
When there is damage to a single strand of DNA, the Poly (ADP-ribose) polymerase (PARP) complex binds to the broken strand to initiate repair. When PARP inhibitors are administered, they trap the PARP complex and DNA replication stalls, causing double strand breaks. According to the synthetic lethality hypothesis, when PARP inhibitors are given to normal cells with wild type BRCA 1 / 2, the cell manages to survive. However, in tumor cells, with mutated BRCA 1 / 2 genes, after administration of PARP inhibitors, there is loss of heterozygosity (LOH), causing synthetic lethality, resulting in the disintegration of the tumor cell (Figure 1).
In advanced PC there is increased frequency of mutations in men with castrate resistant prostate cancer (CRPC). Approximately 21% of men with CRPC have genetic somatic alterations [1], and 11.8% of metastatic PC patients have germline mutations [2]. Moreover, there has been some data demonstrating that approximately 10% of patients with localized PC harbor germline mutations [3], and there might be an association with intraductal/ductal pathology.
Emerging data on the effect of DDR mutation on treatment responses has recently become available. The presence of DDR mutations might be associated with poorer outcomes in patients treated with abiraterone/enzalutamide.
The difference between somatic (sporadic) and germline (inherited) mutations are illustrated in Table 1.

It is important to understand when we should test for these mutations. There are several indications that warrant testing:
1. Known familial mutation in a cancer susceptibility gene
2. Familial history suggestive of hereditary PC syndrome, hereditary breast and ovarian cancer syndrome, or Lynch syndrome
3. Tumor (somatic) sequencing indicating presence of mutations in hereditary cancer risk genes (e.g. BRCA2, BRCA1, ATM, MSH2, MSH6, MLH1, PMS2)
4. According to the NCCN guidelines for genetic testing in 2018, testing should be performed in all metastatic PC patients
5. It remains debatable whether we should test PC patients with high risk localized disease
6. Currently there is no consensus for routine testing, nor for the type of specimen to be tested.
Dr. Pezaro described the TOPARP study [4]. This is a phase 2 trial involving men with metastatic CRPC, with disease progression after 1-2 lines of chemotherapy. Over 80% of men in this trial had more than 4 lines of treatment. Patients received the PARP inhibitor Olaparib 400 mg BD. The primary endpoint was radiographic response (according to RECIST criteria), PSA decline over 50%, and confirmed reduction in the circulating tumor-cell count from 5 or more cells per 7.5 ml of blood to less than 5 cells per 7.5 ml. Overall, 49 patients were enrolled. Sixteen of 49 patients who could be evaluated, demonstrated a response (33%; 95% confidence interval, 20 to 48). The most common adverse effects were anemia (in 10 of the 50 patients [20%]) and fatigue (in 6 [12%]).
There are currently many single agent PARP inhibitors trials in progress (Table 2).

Additionally, there are ongoing combination trials, combining PARP inhibitors with Abiraterone, chemotherapy and immune checkpoint inhibitors (Table 3).

Dr. Pezaro summarized her excellent talk stating that DDR defects are present in a significant minority of men with PC, which are associated with poorer outcomes. Germline and somatic mutation testing offer important data. Germline mutations cannot be predicted by clinical features, and lastly, clinical efficacy data on the usage of PARP inhibitors are emerging. There is much to learn about the choice of agent, and whether combinations are better than single agents. Phase 3 trials are needed to demonstrate evidence of benefit.
Presented by: Carmel Pezaro, Eastern Health and Monash University, Melbourne, Australia
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, Twitter:@GoldbergHanan at the 2018 ASCO Annual Meeting - June 1-5, 2018 – Chicago, IL USA.
References:
1. Robinson et al. Cell 2015
2. Pritchard et al. NEJM 2016
3. Frazer et al. Nature 2017
4. Mateo J. et al. NEJM 2015

