(UroToday.com) The 2024 Advanced Prostate Cancer Consensus Conference (APCCC) meeting featured a session on the treatment for biochemical recurrence/PSA persistence, and a presentation by Dr. Derya Tilki discussing how to manage patients with PSA persistence following radical prostatectomy. Dr. Tilki notes that between 5% and 30% of men continue to have detectable or persistent PSA after radical prostatectomy, which may result from undetected micrometastatic disease, malignant residual disease in the surgical bed, or residual benign tissue at the surgical margins. In the majority of studies, persistent PSA is defined as a detectable post-radical prostatectomy PSA of >0.1 ng/mL within 4-8 weeks of surgery:
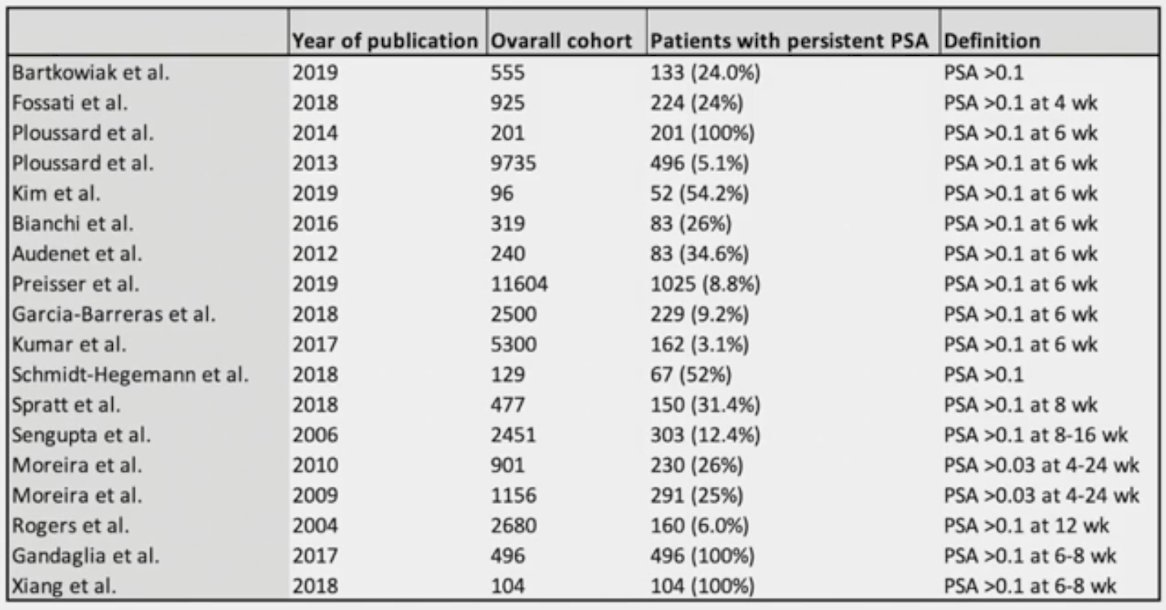
Dr. Tilki notes that PSMA PET/CT imaging can guide salvage treatment strategies. A study from Meijer et al.1 assessed 150 patients with persistent PSA, with a median PSA of 0.60 ng/mL (IQR 0.3-2.4) at the time of PSMA PET/CT. They found that 67% of patients had PSMA positive lesions, including 59% that had lesions outside the prostate fossa and 39% with evidence of distant metastasis:
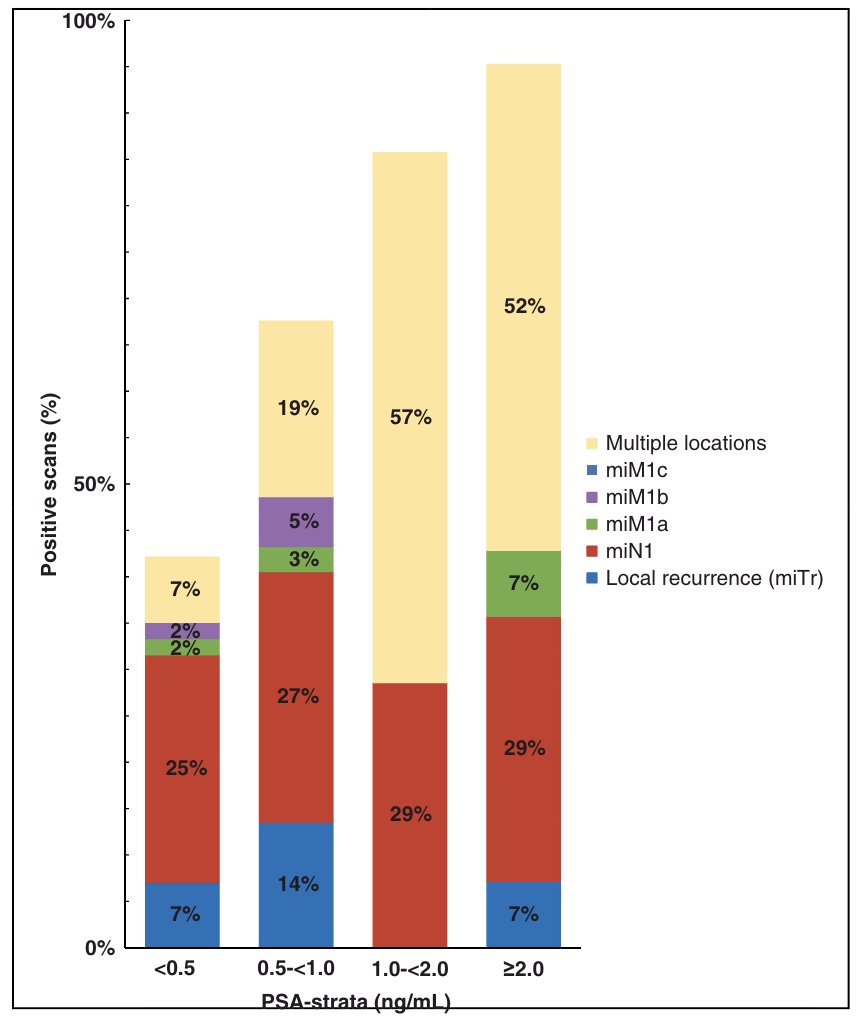
The EAU guideline recommendation for the management of persistent PSA after radical prostatectomy is to offer a PSMA PET/CT scan to men with a PSA > 0.2 ng/mL if the results will influence subsequent treatment decisions (Strength rating: Weak). In 2019, Preisser and colleagues2 assessed the impact of PSA persistence after radical prostatectomy on oncologic outcomes among 11,604 patients undergoing surgery from 1992-2016. Among these patients, 8.8% (n = 1,025) harbored persistent PSA (>= 0.1 ng/mL), and at 15 years after radical prostatectomy, metastasis free survival was 53.0% versus 93.2% (p < 0.001) for persistent versus undetectable PSA:
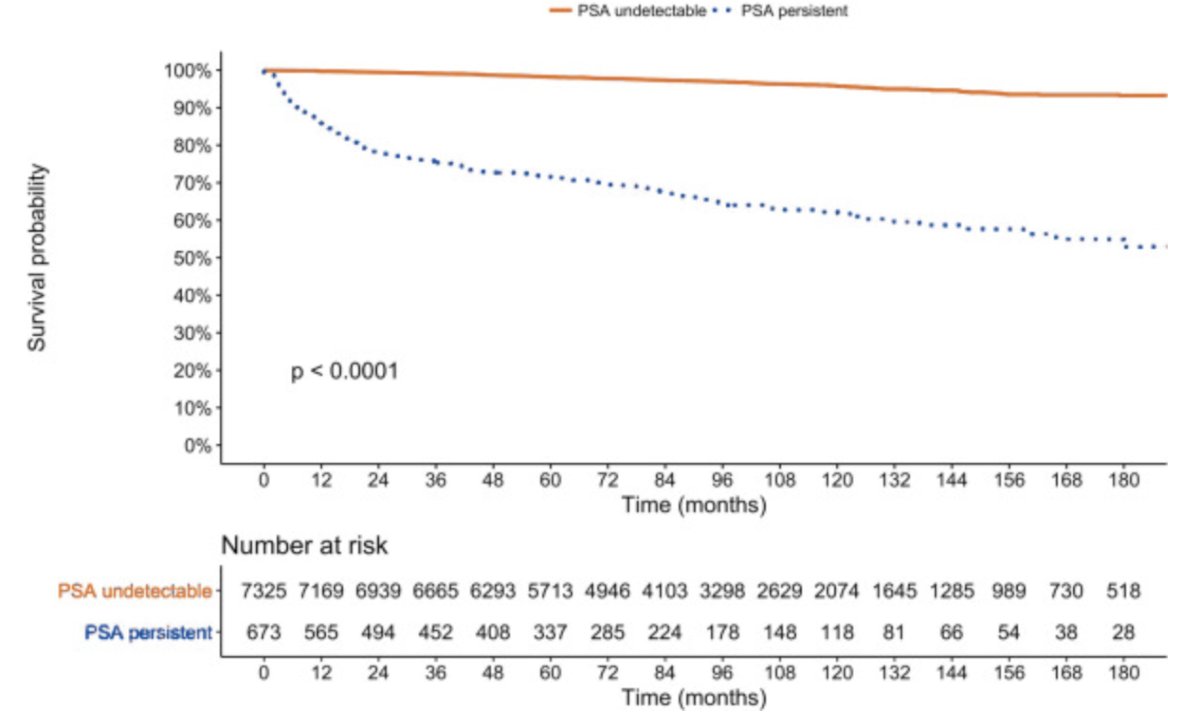
Additionally, in a multivariable analysis from this study, higher PSA, more advanced pathologic tumor stage, pathologic Gleason Group 3-5, positive surgical margins, and pN1 were associated with an increased risk for persistent PSA (all p < 0.01). Moreover, persistent PSA post-operatively was associated with metastasis (HR 3.59, 95% CI 2.83-4.57), death (HR 1.86, 95% CI 1.41-2.45), and cancer-specific death (HR 3.15, 95% CI 1.92-5.18).
Dr. Tilki notes that benign glands are found in ~11% of inked surgical margins, most commonly at the prostate apex, but is likely a rare cause of an elevated PSA after radical prostatectomy.
In a systematic review assessing the management of PSA persistence after radical prostatectomy, Ploussard and colleagues3 screened 2,374 articles, including 25 studies and 5,217 men. PSA persistence was significantly correlated with disease aggressiveness and associated with worse oncologic outcomes than in men with undetectable PSA levels. The 5-year recurrence free survival rates varied from 21.5% to 67.0%.
In 2018, Spratt et al.4 assessed a multi-institutional cohort of 477 men (1990-2015 at three academic medical centers) to assess the impact of the Decipher Genomic Classifier on the risk of metastasis among men with PSA persistence after radical prostatectomy. Patients were classified as detectable PSA (n = 150), or undetectable PSA (n = 327) based on a post-radical prostatectomy PSA nadir > 0.1 ng/mL. On multivariable analysis, only genomic high-risk (HR 5.95, 95% CI 2.02-19.41, p = 0.001), detectable PSA (HR 4.26, 95% CI 1.16-21.8, p = 0.03), and lymph node invasion (HR 12.2, 95% CI 2.46-70.7, p = 0.003) remained prognostic factors for metastasis. Among detectable PSA patients, the 5-year metastasis rate was 0.90% for genomic low/intermediate and 18% for genomic high risk (p < 0.001):
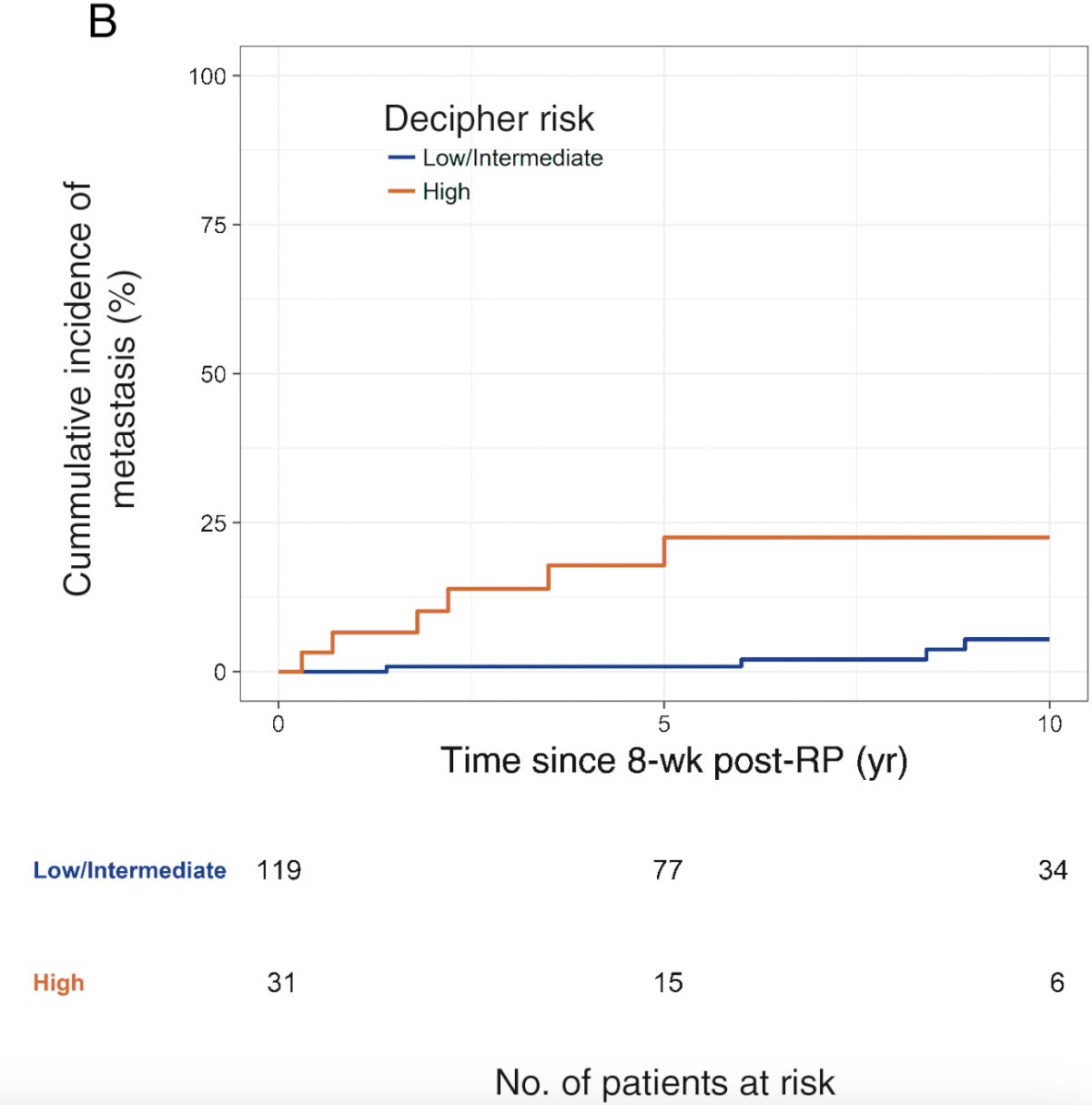
Genomic high risk remained independently prognostic on multivariable analysis (HR 5.61, 95% CI 1.48-22.7, p = 0.01) among detectable PSA patients.
The GETUG-AFU22 trial assessed salvage radiotherapy +/- ADT in patients with PSA persistence, with the following trial design:
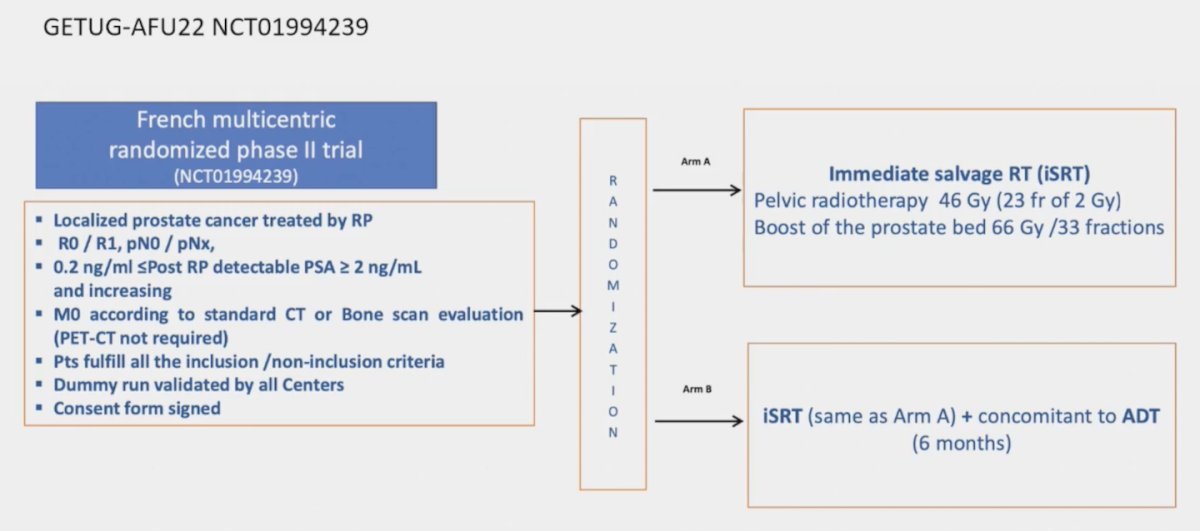
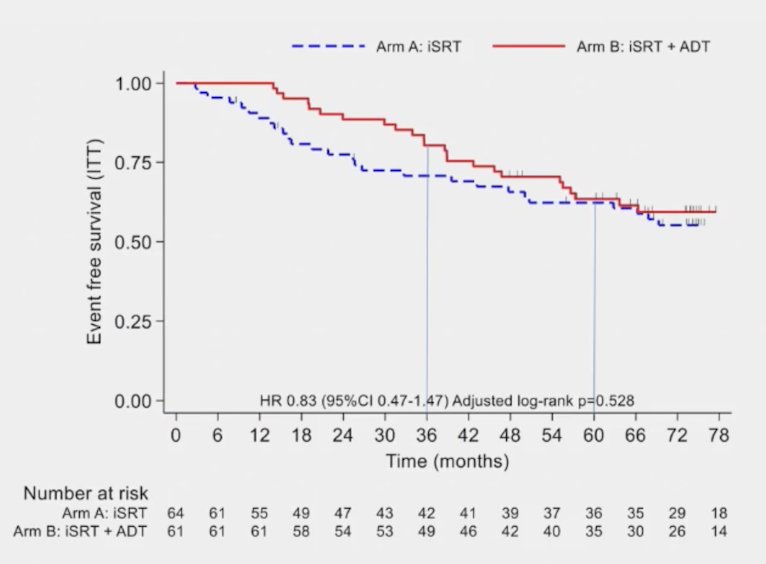
Results were presented at ASTRO 2022 (not peer review published as of yet), noting that at 5 years, the event free survival rate was 62.3% in Arm A and 63.4% in Arm B (HR 0.83, 95% CI 0.47-1.47). The median event free survival was not reached in both arms. With 53 events, the probability of Arm B being better than Arm A is 79%:
Dr. Tilki highlighted that the EAU guidelines also suggest that among men with PSA persistence after radical prostatectomy, we should treat these patients with no evidence of metastatic disease with salvage radiotherapy and additional hormonal therapy (Strength rating: Weak). Finally, Dr. Tilki highlighted several ongoing clinical trials among men with PSA persistence after radical prostatectomy:

Dr. Tilki concluded her presentation by discussing how to manage patients with PSA persistence following radical prostatectomy with the following take-home messages:
- PSA persistence is strongly and independently correlated with adverse pathologic features and poorer survival outcomes
- Risk stratification according to pathologic features, PSA levels/kinetics, and genomic classifier may aid in personalization of treatment
- The current evidence for guiding the management of these patients is still low
- Patients with persistent PSA need to be monitored more closely and treated more aggressively than patients with biochemical recurrence after an undetectable PSA
- Trials of treatment intensification are ongoing
Presented by: Derya Tilki, MD, Martini-Klinik Prostate Cancer Center, University Hospital Hamburg Eppendorf, Hamburg, Germany
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Associate Professor of Urology, Georgia Cancer Center, Wellstar MCG Health, @zklaassen_md on Twitter during the 2024 Advanced Prostate Cancer Consensus Conference, Lugano, Switzerland, April 25th – April 27th, 2024
References:
- Meijer D, Donswijk ML, Bodar YJL, et al. Biochemical persistence of prostate-specific antigen after robot-assisted laparoscopic radical prostatectomy: Tumor localizations using PSMA PET/CT imaging. J Nucl Med. 2021 Jul 1;62(7):961-967.
- Preisser F, Chun FKH, Pompe RS, et al. Persistent Prostate-Specific Antigen After Radical Prostatectomy and its Impact on Oncologic Outcomes. Eur Urol. 2019 Jul;76(1):106-114.
- Ploussard G, Fossati N, Wiegel T, et al. Management of Persistently Elevated Prostate-specific antigen after Radical Prostatectomy: A Systematic Review of the Literature. Eur Urol Oncol. 2021 Apr;4(2):150-169.
- Spratt DE, Dai DLY, Den RB, et al. Performance of a Prostate Cancer Genomic Classifier in Predicting Metastasis in Men with Prostate-specific Antigen Persistence Postprostatectomy. Eur Urol. 2018 Jul;74(1):107-114.
Related Content:

