(UroToday.com) The 2022 Advanced Prostate Cancer Consensus Conference (APCCC) Hybrid Meeting included a session on the management of metastatic hormone sensitive prostate cancer (mHSPC) and a presentation by Dr. Christopher Sweeney discussing treatment intensification strategies in mHSPC, specifically triplet therapies. Dr. Sweeney started by highlighting that there are several life-prolonging treatment options for men with mHSPC.
First, weak ADT with testosterone suppression is better than no treatment at all, specifically testosterone suppression + weak NSAA is better than testosterone suppression alone, which is better than 150 mg of bicalutamide. Second, potent ADT is better than weak ADT. Specifically, extragonadal androgen synthesis inhibition (abiraterone) + testosterone suppression is better than testosterone suppression alone, and potent androgen receptor inhibition (apalutamide, enzalutamide, or darolutamide with docetaxel) + testosterone suppression is better than testosterone suppression. Third, docetaxel with testosterone suppression is better than testosterone suppression alone. Docetaxel + ADT has the strongest and most consistent benefit in patients with de novo high volume disease. Dr. Sweeney notes that for mHSPC patients, there has been a deluge of new data from 2014-2022, whereby we are now up to triplet therapy for appropriate men:

Given that there are multiple life-prolonging treatment options for men with mHSPC, is the overall survival benefit large enough to overcome the impact of sequential treatment for mCRPC, since some patients are not able to receive subsequent therapy secondary to clinical deterioration? Furthermore, we do not yet have clear indication as to which patients benefit from a given regimen, and thus we need to choose a treatment plan for each individual patient with mHSPC.
Going back to the basic oncological principles, the most successful strategies for developing anti-cancer combination therapies commonly adhere to the following:
- Distinct mechanisms of action
- Significant single agent toxicity
- Non-overlapping toxicities
Dr. Sweeney notes that there are significant overall survival benefits by distinct subgroups. First, de novo high volume disease has improved survival for docetaxel + testosterone suppression versus testosterone suppression alone. This benefit has been seen (“consistent”) across 3 of 3 trials, including CHAARTED [1], GETUG15 [2], and STAMPEDE [3], with additional data coming from the STOPCaP IPD scheduled to be presented at ASCO 2022. Second, de novo high volume disease has improved survival for testosterone suppression + NHL versus testosterone suppression alone, as summarized in the following table:

Third, de novo high volume disease has improved survival for ADT + docetaxel + NHT versus ADT + docetaxel, based on data from the PEACE-1 [4] (abiraterone) and ARASENS [5] (darolutamide) trials:
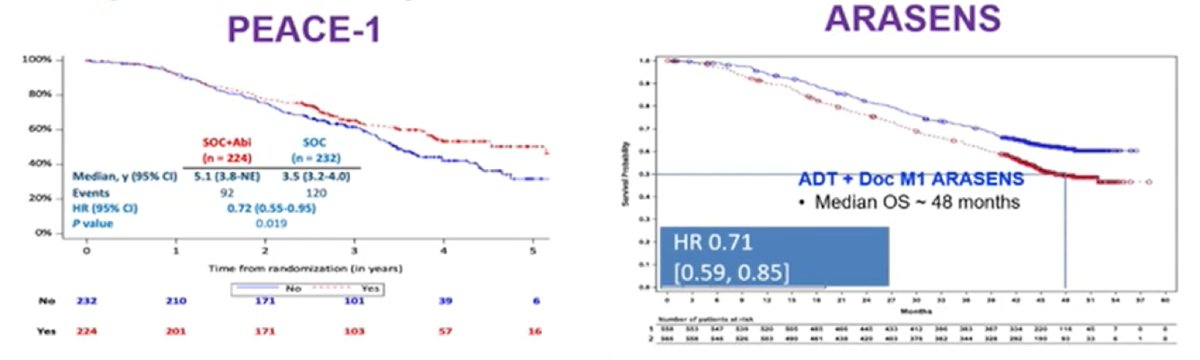
However, there is no data for testosterone suppression + NHT + docetaxel versus testosterone suppression + NHT. In the de novo high volume disease space, we are accumulating data as highlighted by the median OS by treatment (with the addition of ENZAMET after ASCO 2022):

Fourth, de novo low volume disease has modestly improved survival for docetaxel + testosterone suppression versus testosterone suppression alone. This (“modest”) benefit (HR for OS 0.86) has been seen shown across CHAARTED,1 GETUG15,2 and STAMPEDE,3 with again additional data coming from the STOPCaP IPD scheduled to be presented at ASCO 2022. Fifth, de novo low volume disease has improved survival for testosterone suppression + prostate radiotherapy to the primary versus testosterone suppression alone. This is based on data from HORRAD4 and STAMPEDE,5 which showed a significant OS benefit for patients with 4 or less bone metastases.6 Dr. Sweeney notes that there was a more consistent and strong effect and less treatment burden for these patients compared to docetaxel.
There are also certain subgroups that have no clear overall survival benefit. De novo low volume disease testosterone suppression + docetaxel + abiraterone is comparable to testosterone suppression + docetaxel. Data is pending for PEACE-1 to assess testosterone suppression + prostate radiotherapy + abiraterone versus testosterone suppression + prostate radiotherapy. However, there is no data for testosterone suppression + NHT + prostate radiotherapy versus testosterone suppression + NHT, nor for testosterone suppression + NHT + docetaxel versus testosterone suppression + NHT.
There is a possible overall survival benefit for metachronous high volume disease, although the numbers are small:

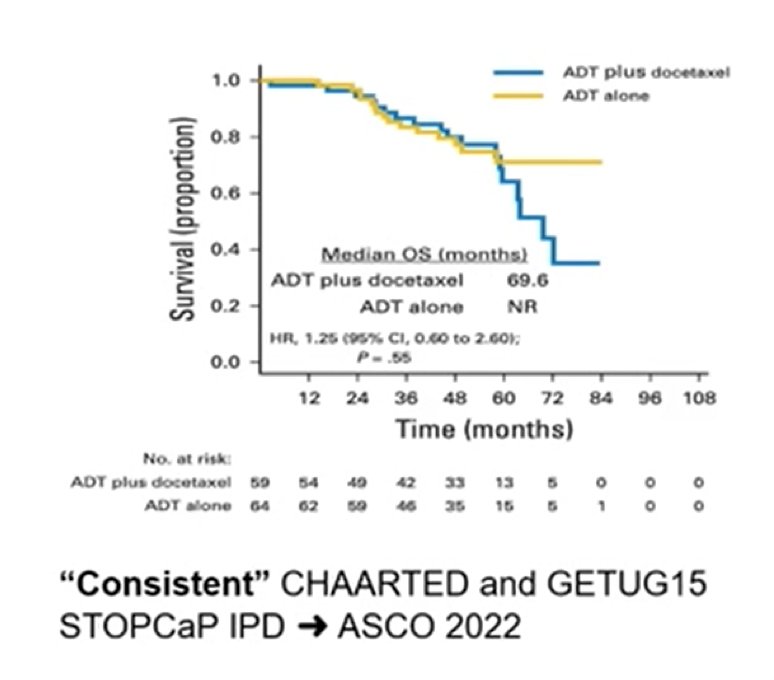
For metachronous low volume disease, testosterone suppression + docetaxel is the same as testosterone suppression, with no additional benefit of docetaxel:
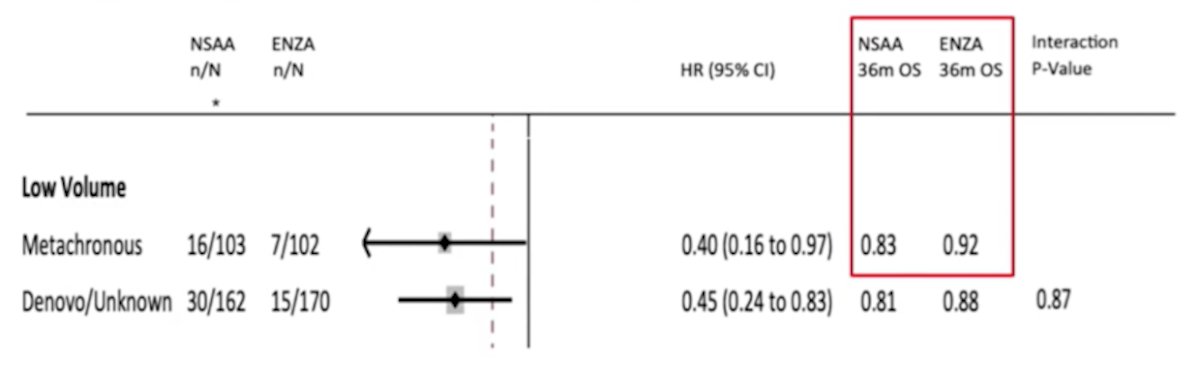
For metachronous low volume disease, testosterone suppression + enzalutamide has an overall survival benefit compared to testosterone suppression + NSAA:
Dr. Sweeney emphasized that at this point in time there are several important points that we do not yet understand:
- The potent hormonal therapy doublet versus potent chemohormonal therapy triplet (testosterone suppression + NHT vs testosterone suppression + NHT + docetaxel). Perhaps we can learn from ENZAMET and/or the planned STOPCaP individual patient data meta-analysis of all the NHT data, but we do not know if this trial will be done. If triplet testosterone suppression + docetaxel + NHT is better than testosterone suppression + NHT, which patients should receive this regimen?
- Testosterone suppression + NHT SBR for oligoprogression vs testosterone suppression + NHT + SBRT vs SBRT testosterone suppression + NHT. This needs to be addressed in patients with metachronous low volume disease, and the question remains as to whether patients should have a treatment break if there is a deep remission
Dr. Sweeney concluded this high-level summary of treatment intensification strategies in mHSPC with the following table highlighting his treatment algorithm based on disease risk groups:

Presented By: Christopher Sweeney, MBBS, Dana-Farber Cancer Institute, Harvard Medical School, Boston, MA
Written By: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 Advanced Prostate Cancer Consensus Conference (APCCC) Annual Hybrid Meeting, Lugano, Switzerland, Thurs, Apr 28 – Sat, Apr 30, 2022.
References:
- Sweeney CJ, Chen YH, Carducci M, et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. N Engl J Med. 2015;373(8):737-746.
- Gravis G, Boher JM, Joly F, et al. Androgen Deprivation Therapy (ADT) Plus Docetaxel Versus ADT Alone in Metastatic Non castrate Prostate Cancer: Impact of Metastatic Burden and Long-term Survival Analysis of the Randomized Phase 3 GETUG-AFU15 Trial. Eur Urol. 2016;70(2):256-262.
- James ND, Sydes MR, Clarke NW, et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet. 2016;387(10024):1163-1177.
- Boeve LMS, Hulshof MCCM, Vis AN, et al. Effect on Survival of Androgen Deprivation Therapy Alone Compared to Androgen Deprivation Therapy Combined with Concurrent Radiation Therapy to the Prostate in Patients with Primary Bone Metastatic Prostate Cancer in a Prospective Randomised Clinical Trial: Data from the HORRAD Trial. Eur Urol. 2019 Mar;75(3):410-418.
- Parker CC, James ND, Brawley CD, et al. Radiotherapy to the primary tumour for newly diagnosed, metastatic prostate cancer (STAMPEDE): A randomized controlled phase 3 trial. Lancet 2018 Dec 1;392(10162):2353-2366.
- Ali A, Hoyle A, Haran AM, et al. Association of bone metastatic burden with survival benefit from prostate radiotherapy in patients with newly diagnosed metastatic prostate cancer: A secondary analysis of a randomized clinical trial. JAMA Oncol. 2021 Apr 1;7(4):555-563.

