The discussion began on the STAMPEDE biomedical imaging group (BIG) and STAMPEDE biomedical research group (BRG) which have incorporated 5,000 CT and bone scans with a target of more than 10,000. The scans are linked to outcome and pathology and annotated for metastasis type, location, and size. One of the reasons for collecting this data is because the bone scan has been shown to be predictive of response to radiotherapy to the primary site when the bone metastasis number is 4 or less. Therefore, at low-volume disease burden, there appears to be a volume threshold effect whereby treatment of the primary with radiotherapy is beneficial.
Next, Dr. Clarke moved on to discuss the various disease volume/risk definitions (Table 1). Unfortunately, none are validated, but all of them agree that non-regional lymph node disease is regarded as low volume disease while visceral disease is regarded as high-volume disease. When assessing the LATITUDE study, examining the role of abiraterone + prednisone + androgen deprivation therapy (ADT) in hormone-sensitive metastatic prostate cancer (mHSPC) patients compared to ADT alone, there was a clear overall survival benefit for the abiraterone treated patients.1 However, when stratifying by disease volume, the benefit was significantly more substantial for the high-volume disease. In the CHAARTED study the docetaxel benefit for high-volume disease was substantially higher than for low-volume disease in patients who presented with de novo metastatic disease. This difference was not apparent in patients with prior local therapy.
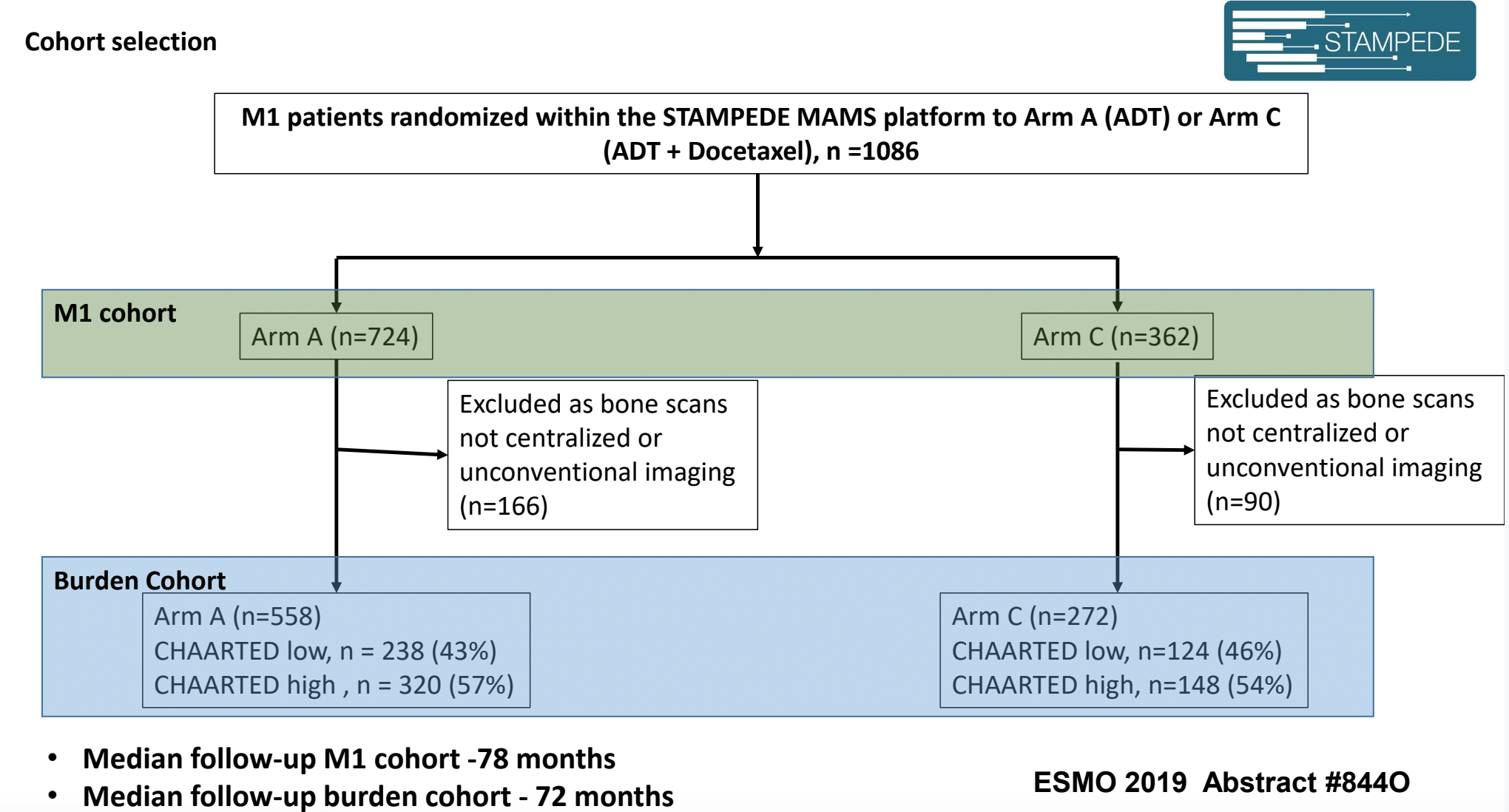
In an abstract that will be presented at ESMO 2019 meeting (#8440), the effect of docetaxel in hormone-sensitive metastatic prostate cancer (mHSPC) in the STAMPEDE trial will be assessed, including a subgroup analysis by metastatic burden. The cohort selection for this study is shown in Figure 1. The results showed that there was no benefit for docetaxel in overall survival for low-burden metastatic disease. Therefore, there is no dichotomized volume/burden related effect relating to docetaxel.
Dr. Clarke concluded his brief talk with two important conclusions:
- At low disease burden, there is a volume threshold effect whereby treatment of the primary tumor with radiotherapy is beneficial. This threshold is predicted by the conventional bone scan.
- Prostate cancer patients presenting with de novo mHSPC should all be considered for combined systemic treatment with docetaxel or novel ADT combined with standard of care ADT.
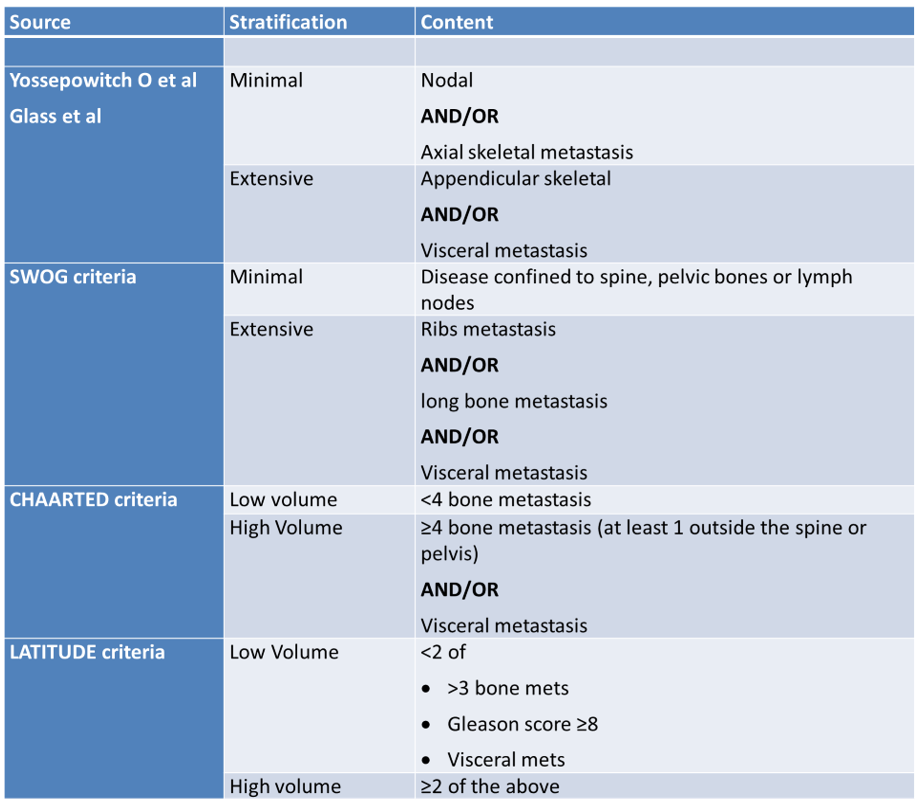
Figure 1 – Cohort selection for abstract 8440 at ESMO 2019 assessing the effect of docetaxel in mHSPC patients in the STAMPEDE trial:
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New York, USA, Twitter: @GoldbergHanan, at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 - 31, 2019 in Basel, Switzerland
Reference:
Further Related Content: Optimal Treatment of Newly Diagnosed of Metastatic Prostate Cancer Presentation

