(UroToday.com) The ANZUP annual scientific meeting’s clinical trials session included an update in germ cell tumors provided by Dr. Ben Tran. Specifically, this included an update on 5 ongoing trials/projects that ANZUP is involved with. The first trial discussed by Dr. Tran was the P3BEP trial, a randomized phase 3 trial of accelerated versus standard BEP chemotherapy for intermediate and poor risk metastatic germ cell tumors. Male and female participants 11-45 years of age with intermediate or poor-risk metastatic germ cell tumor are randomized to Arm A (standard BEP 4 x 21 day cycles) versus Arm B (accelerated BEP 4 x 14 day cycles). End of chemotherapy treatment and safety assessment is performed at 30-42 days after the last study dose, followed by surgical resection of any residual disease +/- further chemotherapy. The final response assessment occurs at 6 months from randomization or after all post-chemotherapy surgery and other interventions are complete. The trial schema for P3BEP is as follows:

As of September 2021, 173 patients have been recruited, supported by a Cancer Australia project grant. Interim analysis results are expected soon, as well as the development of a translational sub-study protocol.
The next trial discussed by Dr. Tran was the TIGER trial, a randomized phase 3 trial comparing conventional-dose chemotherapy using TIP with high dose chemotherapy versus TICE as first salvage in relapsed/refractory germ cell tumors. This is an international trial with a target of 420 patients, including four ANZUP sites (Lifehouse, Princess Alexandra, Box Hill, and the Peter MacCallum Cancer Center). The trial schema for TIGER is as follows:

Multiple previous studies have established that two miRNA clusters are common in testicular germ cell tumors: miR-371-373 and miR-302, with miR-371a-3p performing best. miRNA is present in both adult and pediatric germ cell tumors, including both seminoma and nonseminoma, and they are more sensitive and specific than existing tumor markers. The clinical utility of miRNA is being studied in the SWOG1823 and AGCT1531 clinical trials. The CLIMATE trial is assessing the clinical utility of miR-371 as a marker of residual disease in clinical stage 1 testicular germ cell tumors following orchiectomy. Patients will have miR-371 assessed at baseline, 3, 6, 9, 12, 15, 18, 21, and 24 months, with 200 patients targeted for accrual and a primary endpoint of miR-371 as a marker of residual disease. The trial schema for CLIMATE is as follows:
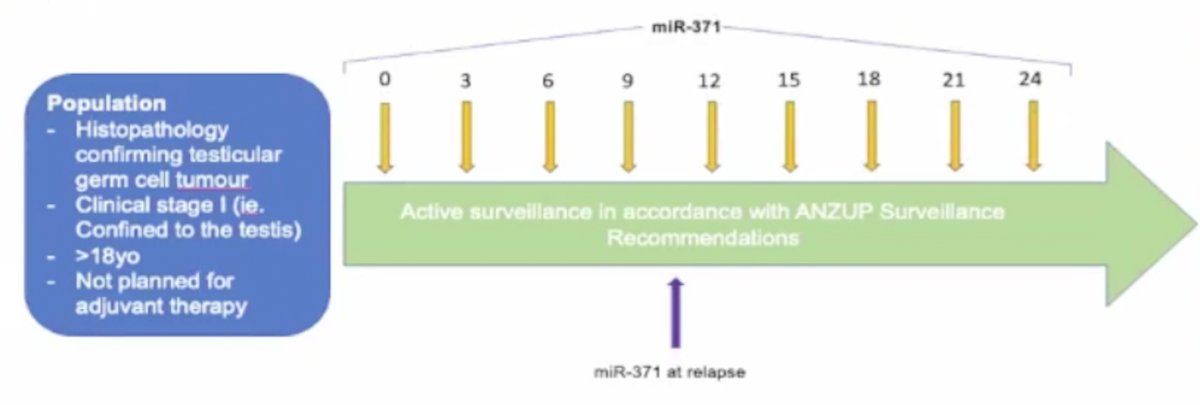
CLIMATE is a parallel study to SWOG1823 and AGCT1531, with an agreement to combine data for a larger analysis.
PRESTIGE is a clinical trial assessing primary retroperitoneal lymph node dissection (RPLND) for clinical stage II testicular germ cell tumor and its impact on health-related quality of life compared to chemotherapy or radiotherapy. There will be 120 patients (either non-seminoma <3 cm lymph nodes, marker negative; or seminoma <3 cm lymph nodes) accrued, targeting 30 patients to primary RPLND and 60 to chemotherapy. The trial schema for PRESTIGE is as follows:

Finally, Dr. Tran discussed iTestis, a testicular cancer registry platform for registry-based trials. Currently, 700 patients are enrolled, with an active surveillance project underway.
Presented by: Ben Tran, MBBS, FRACP, Consultant Medical Oncologist, Peter MacCallum Cancer Centre, Melbourne, Australia
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2021 Australian and New Zealand Urogenital and Prostate (ANZUP) Cancer Trials Group Annual Scientific Meeting (ASM), Sunday, Oct 17 – Monday, Oct 18, 2021.

