(UroToday.com) The 2022 American Urological Association (AUA) Annual Meeting included the International Prostate Forum session and a presentation by Dr. Yaw Nyame discussing the future of PSMA PET/CT in the U.S. Dr. Nyame started his presentation by highlighting the model of prostate cancer clinical disease states.
Conventional imaging (bone scintigraphy, MRI, CT scan) offers sensitivities for detecting lymph nodes ranging from 39-71% and 83-87% for bone lesions. With regards to specificity, lymph node detection is ~82% and for bone detection is 62-90%. Early next generation imaging offered an improvement on detection rates for choline and fluciclovine PET, with detection rates 28-51% for choline PET at PSAs of 0.8-2.0 ng/mL and ~25-60% for PSAs of 0.8-2.0 ng/mL for fluciclovine PET.
Shifting gears to discuss PSMA PET/CT, Dr. Nyame noted that PSMA is a transmembrane protein overexpressed in prostate cancer and that PSMA correlates with aggressive disease. Of note, low levels of PSMA are also expressed in the kidneys, intestines, salivary glands, and brain. The current diagnostic tracers commercially available include 18F-DCFPyL and 68Ga-PSMA-11.
The role of PSMA-PET/CT in the initial staging of prostate cancer was assessed in the proPSMA study.1 This was a multi-center, two-arm randomized controlled trial among men with histologically confirmed prostate cancer who were being considered for curative intent radical prostatectomy or radiotherapy. To be eligible for inclusion, men must have had at least one high-risk factor including PSA greater than or equal to 20 ng/mL, ISUP grade group 3-5, or clinical stage T3 or greater. Following enrollment, patients were randomly assigned in a 1:1 ratio to either conventional imaging performed using bone scan and CT or PSMA PET/CT. The primary study outcome was accuracy of first-line diagnostic imaging for the identification of either pelvic nodal or distant metastatic disease. Between 2017 and 2018, the authors randomly assigned 302 patients of whom 300 received assigned first line imaging. Assessment of the reference standard was possible in 295 (98%) of men, including 87 of whom had evidence of nodal or distant metastasis. In the primary outcome assessment, PSMA PET-CT had a 27% absolute greater AUC for accuracy compared to conventional imaging (95% confidence interval 23-31): 92% (95% CI 88-95%) vs 65% (60-69%). Conventional imaging had both a lower sensitivity (38% vs 85%) and also a lower specificity (91% vs 98%).
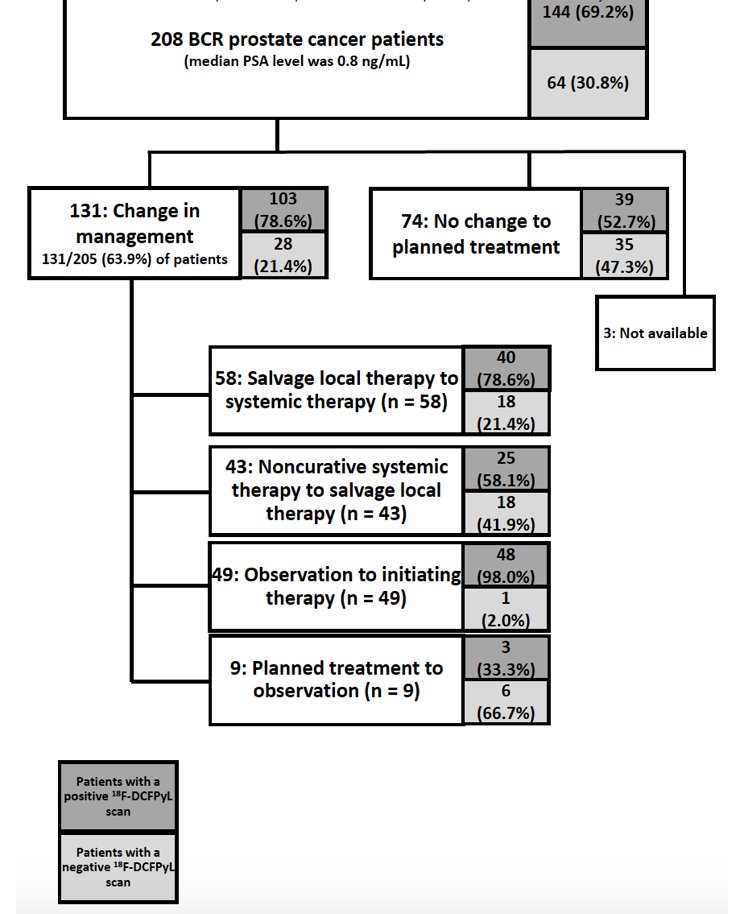
The CONDOR trial focused on patients with biochemically recurrent disease in which the authors recruited men with rising PSA after definitive therapy and negative or equivocal standard of care imaging (e.g., CT/MRI, bone scintigraphy, or F-18 fluciclovine). As their primary outcome of interest, the authors assessed the correct localization rate (CLR), defined as percentage of patients with a 1:1 correspondence between at least one lesion identified by DCFPyL-PET/CT and the composite standard of truth: pathology, correlative imaging, or PSA response, in descending order of priority. CONDOR accrued 208 men who met inclusion criteria, Median PSA in this cohort was 0.8 (range 0.2 - 98.4) ng/mL. Using their defined primary outcome of correct localization rate, the authors demonstrated that DCFPyL-PET/CT correctly localized lesions in 84.8-87.0% of cases among the three readers (lower bound of 95% CI: 77.8%-80.4%), against the composite SOT.
While these imaging characteristics are important, potentially more important is the demonstrated clinical effect of this information. Using local radiographic assessment, PSMA-avid lesion(s) were found in 142 of 208 patients (69.3%). As a result, more than two thirds of patients enrolled in this study (131 of 205, 63.9%) had a change in intended management following DCFPyL-PET/CT. Of those with a change in management, nearly 80% (103/131, 78.6%) were attributable to positive DCFPyL findings while the remaining 21.4% (28/131) were attributable to negative DCFPyL scans. Changes in management included a transition from salvage local therapy to systemic therapy on the basis of more extensive disease (n=58), a period of observation (n=49), non-curative systemic therapy to attempted curative salvage local therapy (n=43) and observation in place of planned treatment (n=9):
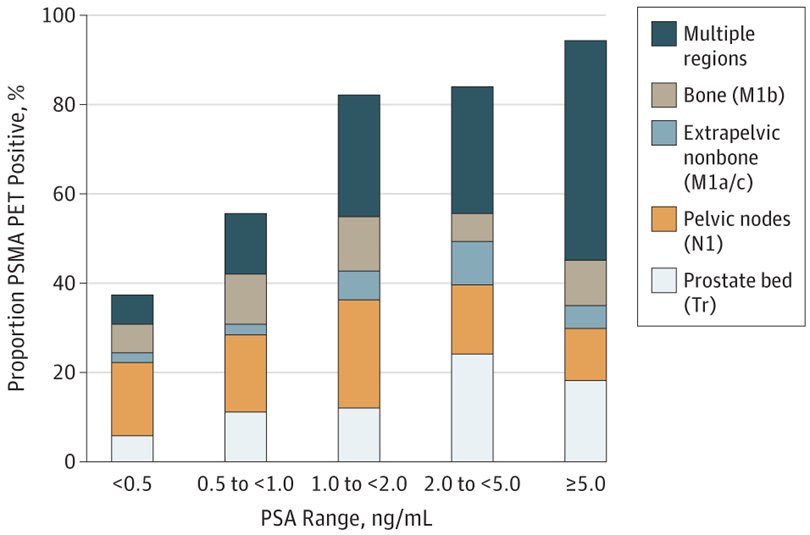
Fendler and colleagues have also assessed 68Ga-PSMA-11 PET accuracy in localizing recurrent prostate cancer in a prospective single-arm trial [3]. This trial included 635 patients with biochemically recurrent prostate cancer after prostatectomy (n = 262, 41%), radiation therapy (n = 169, 27%), or both (n = 204, 32%) that underwent 68Ga-PSMA-11 PET. 68Ga-PSMA-11 PET localized recurrent prostate cancer in 475 of 635 (75%) patients, with detection rates significantly increased with PSA: 38% for <0.5 ng/mL (n = 136), 57% for 0.5 to <1.0 ng/mL (n = 79), 84% for 1.0 to <2.0 ng/mL (n = 89), 86% for 2.0 to <5.0 ng/mL (n = 158), and 97% for ≥5.0 ng/mL (n = 173, p < .001):
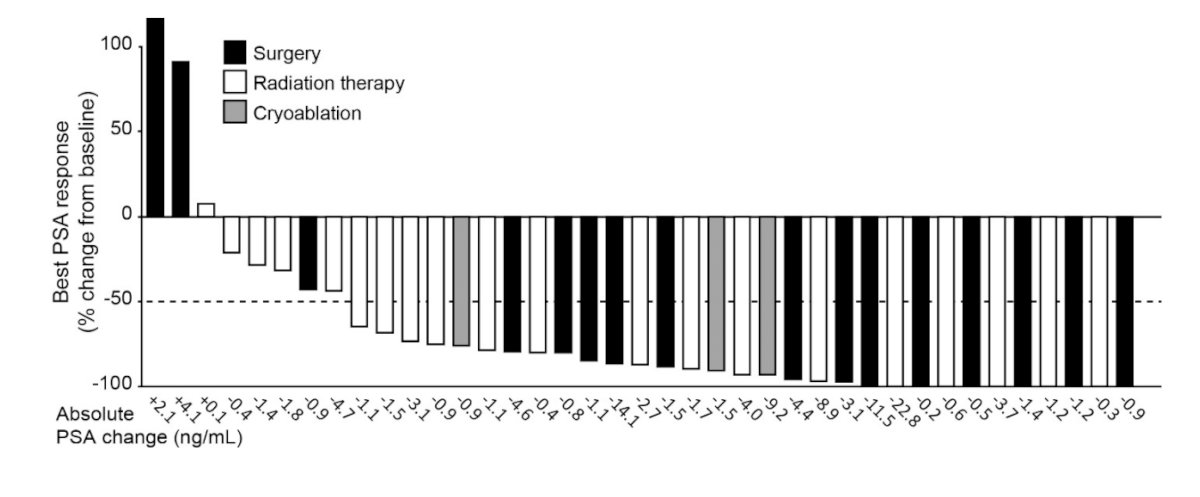
PET-directed focal therapy alone led to a PSA drop of 50% or more in 31 of 39 (80%) patients:
Dr. Nyame then discussed the recent updates to several of the guidelines for next generation imaging. The RADAR III recommendations are as follows:
- Newly diagnosed patients: if conventional imaging is equivocal or negative with continued high suspicion for metastatic disease, consider next generation imaging
- Biochemical recurrent patients: consider next generation imaging for PSA >= 0.5 ng/mL, and PSA < 0.5 ng/mL can be considered based on specific performance of various next generation imaging
- M0 CRPC: only consider next generation imaging in the setting of PSADT < 6 months, when M1 therapies would be appropriate
- M1 CRPC: utilize conventional scans and consider next generation imaging only if conventional scans are negative and the clinician still suspects disease progression. Next-generation imaging should be based on any one of the following: (i) with every doubling of PSA since the previous imaging, (ii) every 6-9 months in the absence of PSA rise (iii) change in symptomatology, and (iv) chance in performance status
The AUA guidelines, recently presented at the AUA 2022 meeting, state that in patients with prostate cancer at high risk for metastatic disease with negative conventional imaging, clinicians may obtain molecular imaging to evaluate for metastases (Expert Opinion). The EAU guidelines sate that PSMA PET/CT is more accurate for staging than CT and bone scan for high-risk disease, but to date no outcome data exist to inform subsequent management. Dr. Nyame emphasized that there are currently several unanswered clinical situations: (i) success and durability of PSMA directed therapies, (ii) incorporating into current treatment paradigms, (iii) new PSMA molecules.
Dr. Nyame concluded his presentation discussing the future of PSMA PET/CT in the U.S. with the following summary points:
- PSMA PET/CT provides greater diagnostic accuracy compared to other imaging modalities
- Diagnostic accuracy of PSMA PET/CT is around 30-40% for men with a PSA < 0.5 ng/mL
- Data are lacking to inform guidelines on PSMA directed therapy
Presented by: Yaw A. Nyame, MD, MS, Department of Urology, University of Washington, Seattle, WA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Urological Association (AUA) Annual Meeting, New Orleans, LA, Fri, May 13 – Mon, May 16, 2022.

