(UroToday.com) In a podium presentation at the 2022 American Urologic Association Annual Meeting held in New Orleans and virtually, Dr. Fleming presented results from the SPOTLIGHT study examining the role of 18F-RHPSMA-7.3 PET in patients with biochemical recurrence (BCR) following local therapy for prostate cancer.
This is a rapidly moving disease space. Accurate localization of disease at the time of BCR is important to guide treatment decisions, informing salvage therapies either locally, systemically, or in combination. Recently, next generation imaging has emerged as having a key role in this disease space, including a large role for prostate-specific membrane antigen (PSMA) -targeted positron emission tomography (PET) radiopharmaceuticals. 18F-rhPSMA-7.3 represents a new class of high affinity PSMA-targeted PET radiopharmaceuticals with the potential for low bladder activity. 18F-rhPSMA-7.3 has particular potential given that 18-F has longer half-life, the ability for larger batch production, and improved spatial resolution compared to 68Ga. Further, the rhPSMA radiohybrid platform allows the potential for labeling with alpha or beta-emitting radiometals for systemic radiation therapy.
The SPOTLIGHT trial (NCT04186845) assesses the diagnostic efficacy of 18F-rhPSMA-7.3 in men with BCR.

In this presentation, Dr. Fleming reports results relating to the impact of 18F-rhPSMA-7.3 on patient upstaging compared with conventional imaging
In SPOTLIGHT, men with suspected BCR underwent PET 50–70 min after IV administration of 296 MBq 18F-rhPSMA-7.3. As a gold standard for comparison, PET results were validated using histopathology and/or conventional imaging as a composite Standard of Truth (SoT). Image guided biopsies of PET lesions took place within 60 days post-PET and confirmatory imaging took place within 90 days. Three blinded central readers evaluated the 18F-rhPSMA-7.3 PET scans. Assessing the question of upstaging, these readers identified the number of positive 18F-rhPSMA-7.3 scans in defined regions that led to upstaging for patients having a documented negative baseline conventional imaging (performed ≤118 days before PET). These were stratified according to the patients’ prior therapy.
The authors included 366 men across 27 US/European sites who underwent 18F-rhPSMA-7.3 PET and had sufficient data to determine a SoT. Among these men, the mean [range] PSA was 3.95 [0.03–134.60] ng/mL at the time of imaging. Overall, 57% of these men had at least one 1 true positive lesion.
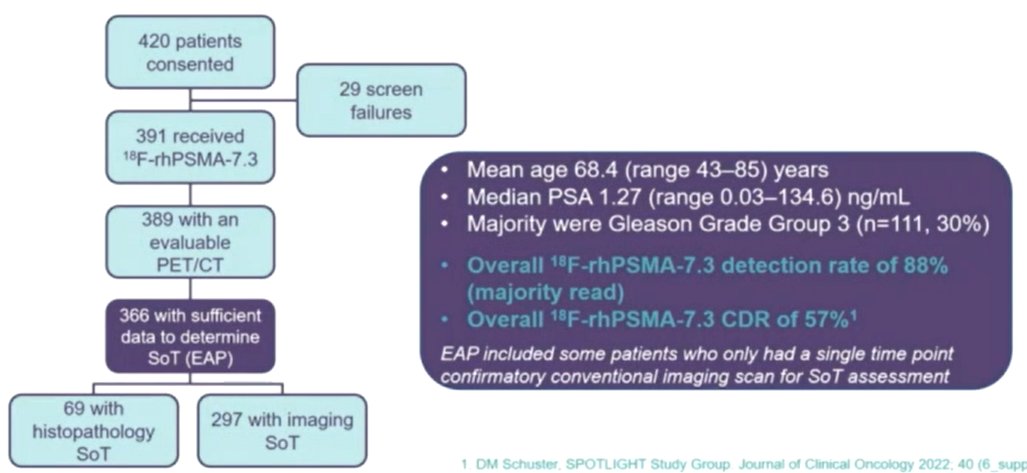
Among these included patients, 250 of 366 (68%) had negative conventional imaging. In this subset, 18F-rhPSMA-7.3 PET had a CDR of 45-47% across three readers, resulting in upstaging compared to conventional imaging. However, there were notable differences in disease distribution on the basis of prior local therapy modality.
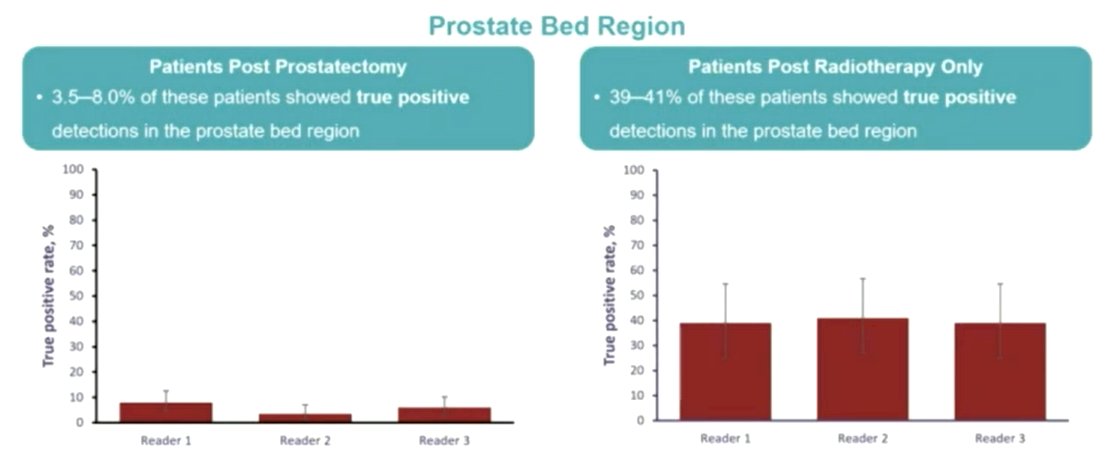
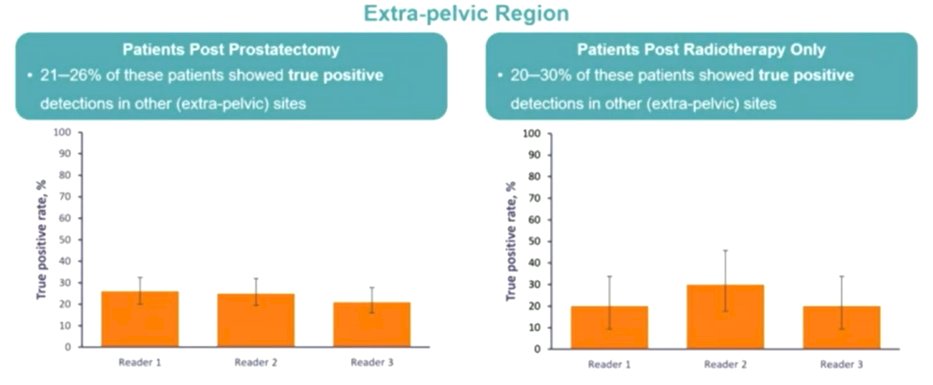
Among patients who had undergone prostatectomy, relatively few 18F-rhPSMA-7.3 positive scans showed lesions in the prostate bed region (3.5-8.0%) with disease found more commonly in pelvic lymph nodes (18-21%) and in other sites (21-26%) that led to upstaging vs conventional imaging.
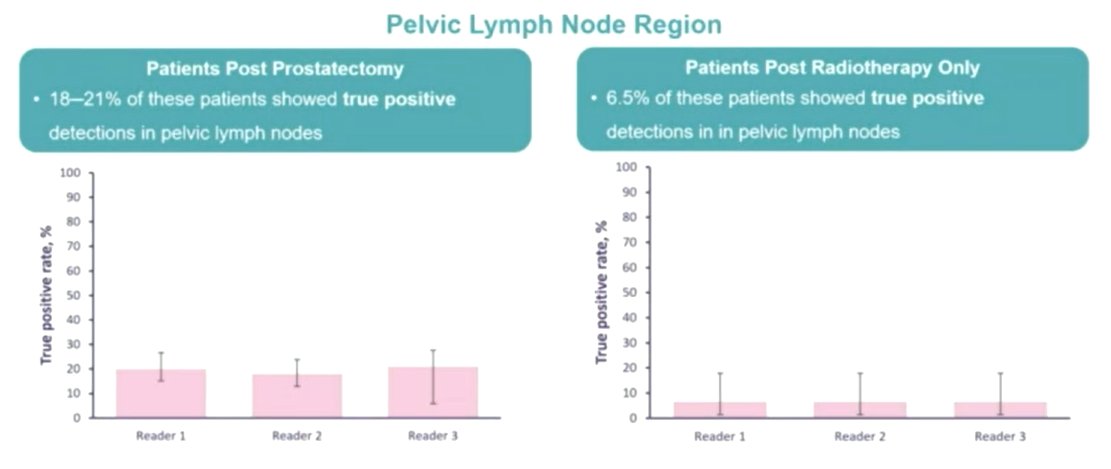
Among those who had received radiotherapy, prostate bed recurrent disease was much more common (39-41%), with recurrence in lymph nodes found in 6.5% and in other sites in 20-30%.
Very few patients had an alternative primary therapy; thus, no definitive conclusions could be drawn for them.
Dr. Fleming then provided a case example in which 18F-rhPSMA-7.3 PET diagnosed extra-pelvic nodal disease which substantially changed patient management.
Dr. Fleming therefore concluded that, at the time of BCR, 18F-rhPSMA-7.3 PET findings frequently resulted in post-scan upstaging compared with conventional imaging, particularly among patients with intact prostates. The use of 18F-rhPSMA-7.3 PET may help to better define sites of disease recurrence and inform salvage therapy decisions.
Presented by: Mark Fleming, MD, Virginia Oncology AssociatesWritten by: Christopher J.D. Wallis, University of Toronto Twitter: @WallisCJD during the 2022 American Urological Association (AUA) Annual Meeting, New Orleans, LA, Fri, May 13 – Mon, May 16, 2022.

