(UroToday.com) Advances in imaging have dramatically reshaped prostate cancer diagnosis, staging, and treatment. While multiparametric MRI (mpMRI) of the prostate has dramatically changed prostate cancer diagnosis, molecularly-targeted imaging has reshaped disease staging and detection of recurrent disease. Conventional imaging approaches using including computed tomography (CT) and bone scan have relatively limited sensitivity to detect disease recurrence following initial local therapy. A number of studies have demonstrated that prostate-specific membrane antigen (PSMA) targeting PET radiopharmaceuticals detect disease not identified based on conventional imaging, with greater accuracy, for initial staging and detection of biochemical recurrence. In a presentation in the Center for Therapy Excellence Young Investigator Award session at the Society of Nuclear Medicine & Molecular Imaging (SNMMI) 2021 Annual Meeting, Dr. Petra Lovrec discussed the potential for prostate-specific membrane antigen (PSMA)-based 18F-DCFPyL (PyL) PET imaging to predict metastatic disease response to therapy and to assess metastatic disease patterns of response and recurrence for patients receiving neoadjuvant chemohormonal therapy followed by prostatectomy for high-risk prostate cancer.
To do so, the authors performed a baseline PyL whole-body PET/CT (PET1) for patients with newly diagnosed high-risk prostate cancer. Patients then received neoadjuvant docetaxel and androgen deprivation therapy. The patient then underwent a second PET/CT (PET2) followed by a radical prostatectomy. Patients underwent a third PET/CT (PET3) at one year following prostatectomy or earlier if PSA progression criteria were met.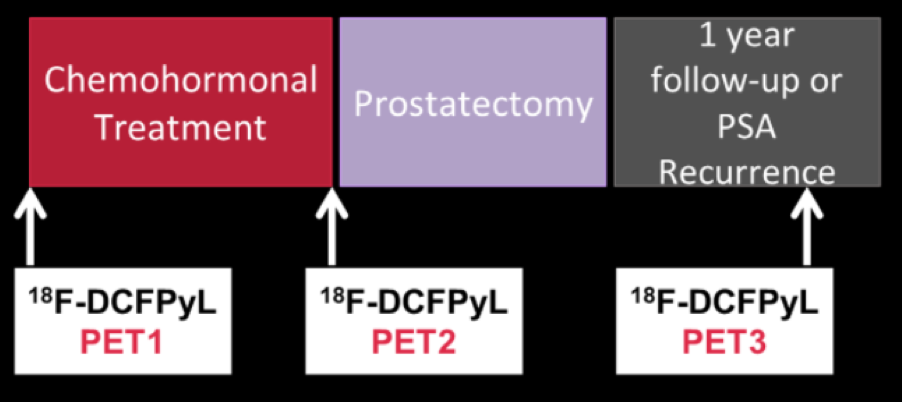
Each PSMA-PET/CT was evaluated by independent-reader central review by two readers with third-reader adjudication. Sites of suspected PET-positive metastatic disease were evaluated by a 5-point scale, anatomic region (pelvic nodal [LN], extrapelvic LN, and bone), and maximum standardized update value (SUVmax). Scores of 3 or greater on a 5-point scale were considered positive. The authors further considered the interval SUVmax (iSUV) change between each time point and operationalized this as absolute and percent change for the highest SUVmax intra-patient metastasis (hottest lesion) and a max of 5 lesions per region (tumor burden).
To understand the clinical importance of these radiologic studies, the PET findings were correlated to progression type based on PSA progression (no progression [NP], early progression ≤1-year post-prostatectomy [EP], or late progression > 1-year post-prostatectomy [LP]), time to progression (TTP) and interval PSA change (iPSA).
The authors included 27 patients of whom, at the time of evaluation, 10 were disease-free, 16 demonstrated early or late recurrence, and 1 was lost to follow-up. At baseline, the median age was 61 years (range 43-70 years) and median PSA was 21 ng/mL (range 4.5-356.7 ng/mL). The median follow-up time was 659 days (range 364-938).
Among the 16 patients with recurrent disease, 12 presented with metastatic disease on initial exam, of whom 10 subsequently exhibited early PSA progression post-prostatectomy.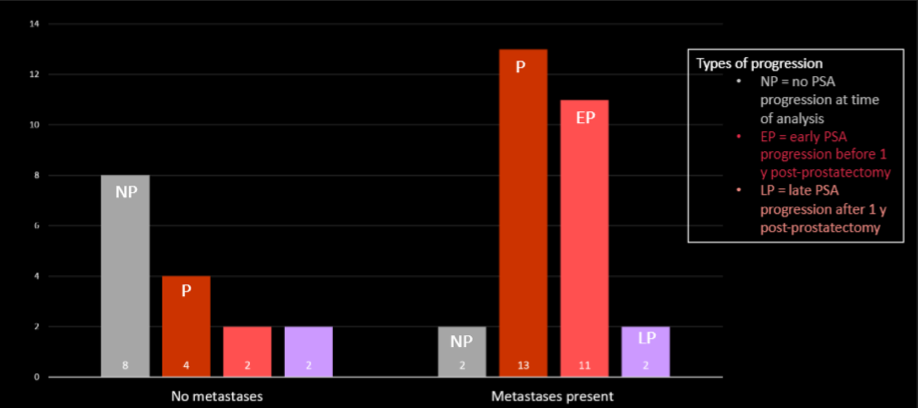
Patients with bone and extrapelvic LN metastases have worse outcomes with evidence of early post-prostatectomy progression in four of 5 with bony disease and seven of seven with extrapelvic lymphadenopathy. Further, all eight patients with more than three pelvic lymph nodes on baseline PSMA-PET/CT had early post-prostatectomy progression. In terms of disease pattern, no new sites of metastatic disease were seen on PET2 or PET3, and all patients progressed at the sites of initially identified metastases on PET1. Among the 10 patients who had no evidence of metastatic disease on baseline scan (PET1), eight had durable PSA response to therapy.
The authors further found that the distribution of disease was significantly associated with the meantime to progression, in particular with an increased risk of progression among those with extrapelvic lymph node metastasis (HR 3.85, p=0.04; compared to no extrapelvic lymph node metastasis) and polymetastatic pelvic disease (HR 1.95, p=0.03; compared to oligometastatic pelvic nodal disease).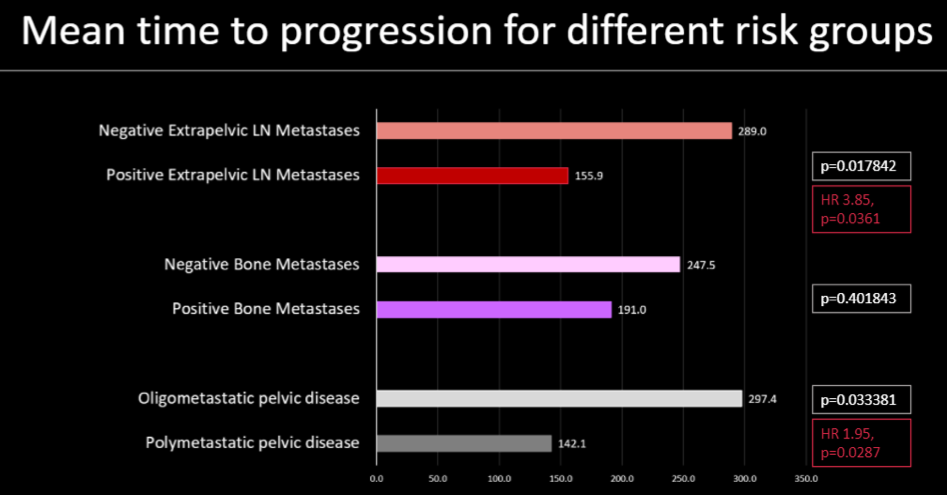
The authors demonstrated a significant correlation between the interval change in tumor burden SUVmax and the interval change in PSA (r =-0.58), but no significant correlation was found between interval change in tumor burden SUVmax change and time to PSA progression (r=-0.25).
Among those with no evidence of progression following radical prostatectomy, the median absolute interval change in tumor burden SUVmax was -0.20 (range -0.10 to -0.30), whereas it was -3.60 (range 2.45 to -25.20) for those with early post-prostatectomy progression and -2.60 (range -0.70 to -4.50) for those with late post-prostatectomy progression. Similarly, the median percent interval change in tumor burden SUVmax for those with no evidence of progression following prostatectomy was -7.70 % (range -3.85 to -11.54), whereas it was -59.16 % (range 13.69 to -84.37) for those with early post-prostatectomy progression and -54.53 % (range -24.14 to 84.91) for those with late post-prostatectomy progression.
The initial hottest lesion SUVmax of the “hottest” or most avid lesion differed significantly between patients with no progression, early progression, and late progression following radical prostatectomy with higher PSMA avidity in the hottest lesions among patients with early progression.
Additionally, SUVmax of the “hottest” or most avid lesion was inversely associated with time to progression (r =-0.48).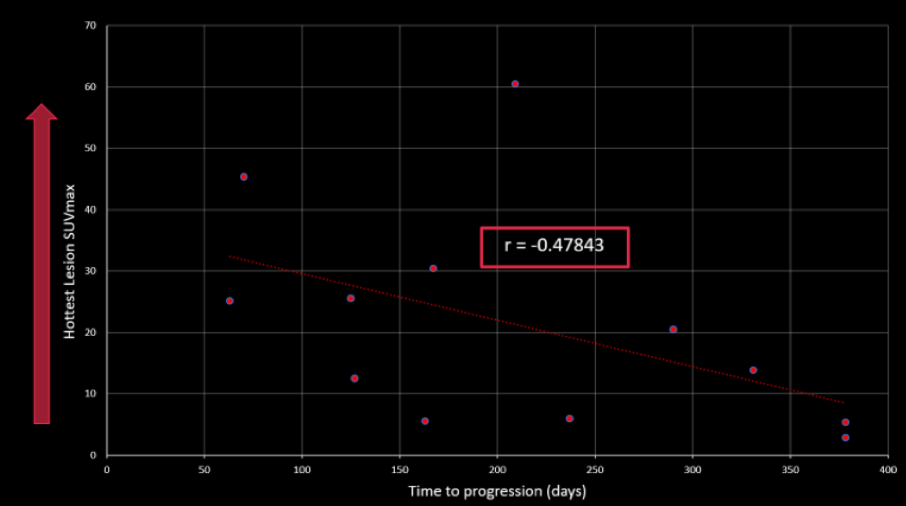
Thus, the authors concluded that results of initial staging with DCFPyL PET/CT prior to neoadjuvant chemohormonal therapy and radical prostatectomy for high-risk prostate cancer were prognostic for post-prostatectomy progression. In particular, detection of more than three intrapelvic lymphadenopathies, any extrapelvic lymphadenopathy, or bone metastases in patients was associated with early progression following radical prostatectomy. Interestingly, at the time of progression, no new sites of PET-positive metastatic disease were found at the time of progression but rather, there was progression at the same sites. Additionally, the PSMA avidity of the hottest lesion was associated with the time to progression.
Presented by: Petra Lovrec, MD, University of Wisconsin-Madison Madison, WI