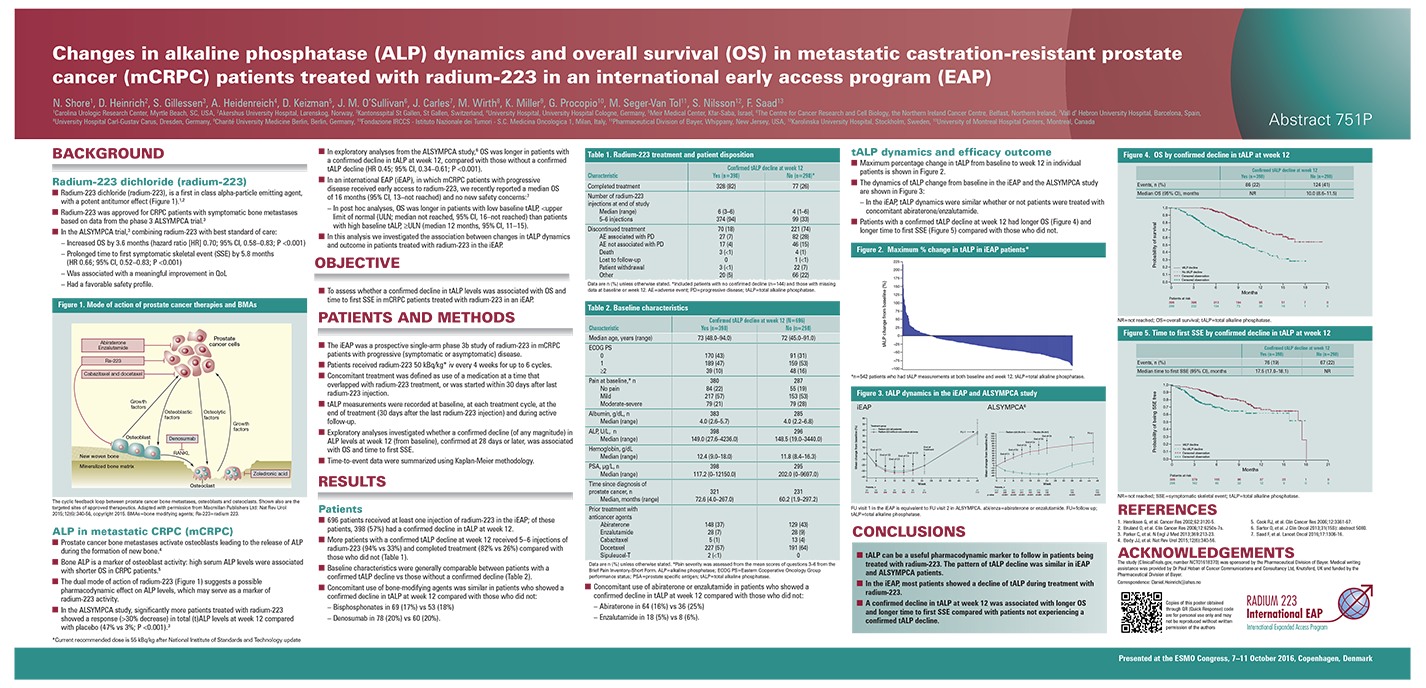
In exploratory analyses of mCRPC pts with bone metastases treated with Ra-223 in the ALSYMPCA trial, OS was significantly longer in patients with a confirmed decline in ALP levels from baseline at week 12, compared with pts without a confirmed ALP decline. Here, we present data on ALP dynamics and OS and time to first symptomatic skeletal events (SSE) in pts treated with Ra-223 in an international EAP.
This was a prospective single-arm phase IIIb study of CRPC patients with symptomatic or asymptomatic bone metastases (no visceral disease) recruited from 14 countries. Patients received Ra-223 50 kBq/kg (55 kBq/kg after NIST update) iv, every 4 weeks for up to 6 cycles. Co-primary endpoints were safety and OS. Exploratory analyses investigated whether a confirmed decline (any magnitude) in ALP levels was associated with OS and time to first SSE.
696 ppatients received at least one Ra-223 cycle. Of those, 398 (57%) patients had a confirmed decline in ALP and 298 (43%) had no confirmed ALP decline. Key baseline characteristics are shown in the poster. More patients with a confirmed ALP decline (374, 94%) received 5–6 Ra-223 injections than those with no ALP decline (99, 33%). Hazard ratios (HR) for confirmed ALP decline at week 12 vs no decline suggest a strong association of ALP decline with both longer OS (HR 0.299, 95% CI 0.227–0.395) and longer time to first SSE (HR 0.474, 95% CI 0.340–0.662) (Poster).
In this early access program (EAP), which is relevant for patients currently treated in clinical practice, decline in ALP was associated with longer OS and time to the first SSE.
Authors:
Neal Shore, Carolina Urologic Research Center, Myrtle Beach, SC, USA; Daniel Heinrich Akershus University Hospital, Lørenskog, Norway; Silke Gillessen, Kantonsspital St Gallen, St Gallen, Switzerland; Axel Heidenreich, University Hospital, University Hospital Cologne, Germany; Daniel Kejzman, Meir Medical Center, Kfar-Saba, Israel; Joe M. O’Sullivan, The Centre for Cancer Research and Cell Biology, Belfast, Northern Ireland; Joan Carles Vall d’ Hebron University Hospital, Vall d’Hebron Institute of Oncology, Barcelona, Spain; Manfred Wirth, University Hospital Carl-Gustav Carus, Dresden, Germany; Kurt Miller, Charité University Medicine Berlin, Berlin, Germany; Giuseppe Procopio, Fondazione IRCCS - Istituto Nazionale dei Tumori - S.C. Medicina Oncologica 1, Milan, Italy; Monica Seger-Van Tol, Pharmaceutical Division of Bayer, Whippany, New Jersey, USA; Sten Nilsson, Karolinska University Hospital, Stockholm, Sweden; Fred Saad, University of Montreal Hospital Centers, Montreal, Canada