However, this has changed with the introduction of the 68Ga-PSMA-11 PET imaging. Due to the overexpression of the prostate-specific membrane antigen (PSMA) on the cell surface of most PC cells, this molecule has been identified as an ideal target for PET imaging using specific tracers that bind to the extracellular domain of PSMA. Level 2b evidence based on superior detection rates after RP, led to a grade A recommendation of PSMA PET/CT by the European Association of Urology (EAU) guidelines.
Nowadays, when BCR develops, even small sub-centimeter metastatic lymph nodes could be visualized in patients with low PSA values. Although, detection of metastatic lesions by 68Ga-PSMA-11 PET imaging still requires a minimal size of the metastatic deposit, and despite the fact that not all PC lesions exhibit PSMA overexpression, growing evidence confirms the superiority of this novel imaging technique.
This has fueled the pursuit of salvage lymph node dissections (sLND) in patients with early BCR with only lymphatic disease demonstrated on imaging. However, a major challenge during sLND is the fact that these metastatic lymph nodes deposits might not be easily detected intraoperatively, due to their inconspicuous morphology and/or atypical localization. This has unfortunately resulted in cumbersome preparation of the tissue and usage of intraoperative frozen section analysis of dissected specimens.
In an attempt to overcome these problems, an intraoperative method has been developed that will enable us to detect the metastatic lesion is desirable. Thus, specific PSMA tracers were manufactured so that they can be used for intraoperative detection of soft tissue metastases. By labeling with gamma-emitting isotopes, PSMA-directed ligands could be developed that would facilitate intraoperative detection during sLND.
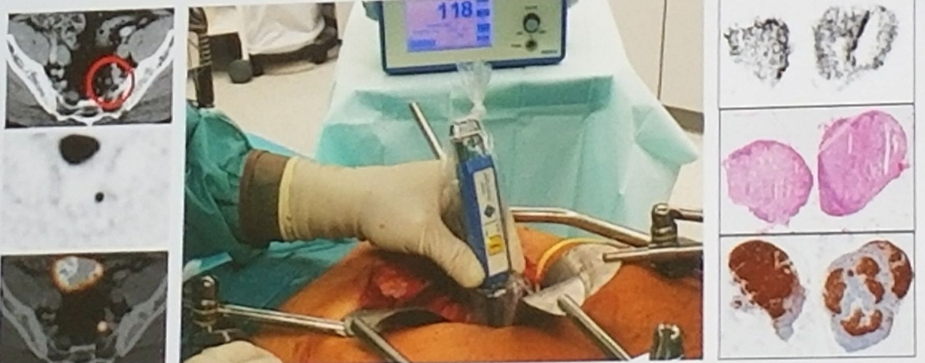
These PSMA-radioguidance 111Indium-labeled PSMA ligands are injected intravenously 24 to 48 h prior surgery. Comparable to 68Ga-PSMA-11, the tracer accumulates not only in the metastatic lesions, but also within the salivary glands, the liver, spleen and small intestines, and is excreted by the kidneys. At the time of surgery, however, the tracer is mostly cleared from circulation and does not exhibit extensive background signal within the fatty tissue where metastatic lymph nodes or soft tissue lesions are located. Thus, radioactive-labeled metastatic PC lesions can be detected by measurements with a sterile-draped handheld gamma probe (Figure 1).
It is important to note, that low-volume small-sized PC lesions might neither be detected by preoperative 68Ga-PSMA-11 PET, nor by intraoperative gamma probe measurements. Thus, Dr. Maurer recommends dissection of surrounding tissue as well. In any case, the templates in these salvage surgery settings, are entirely a matter of debate.
This sLND surgical approach is still completely experimental. Careful selection of suitable patients based on the findings of 68Ga-PSMA-11 PET imaging, clinical variables, as well as the overall medical status, and predicted life expectancy must be taken into consideration. Short- and long-term outcomes must be evaluated, as well as the morbidity and complication rates of this procedure. In the meantime, postoperative PSA-responses can serve as short-term outcomes for successful removal of macroscopic disease as seen on 68Ga-PSMA-11PET.
Figure 1: Radioactive-labeled metastatic prostate canner lesions detected by measurements with a sterile-draped handheld gamma probe:
Speaker: Tobias Maurer, MD Attending Physician Technische Universität München, TUM Urological Clinic Munich, Germany
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre @GoldbergHanan at The 15th Meeting of the EAU Section of Oncological Urology ESOU18 - January 26-28, 2018 - Amsterdam, The Netherlands

