(UroToday.com) The 2023 ESMO annual meeting included a session on prostate cancer, featuring a presentation by Dr. Christian Gratzke discussing results of the phase 3 KEYNOTE-991 trial assessing pembrolizumab + enzalutamide and ADT for patients with metastatic hormone-sensitive prostate cancer (mHSPC).
Indeed, new therapeutic options are needed to delay disease progression for patients with mHSPC. Previously, the combination of pembrolizumab + enzalutamide has shown antitumor activity in patients with metastatic prostate cancer. The phase 3 KEYNOTE-991 study investigated pembrolizumab or placebo + enzalutamide + ADT in patients with next-generation hormonal agent-naïve mHSPC.
Eligible patients (aged ≥18 yrs, ECOG performance status ≤1) had confirmed mHSPC (≥2 bone lesions and/or visceral disease, verified centrally), no prior next-generation hormonal agent, and had completed any prior docetaxel (≤6 cycles) ≤2 months from randomization. Patients were randomized 1:1 to receive pembrolizumab 200 mg or placebo IV every 3 weeks for ≤35 cycles + enzalutamide 160 mg orally daily + continuous ADT (if no history of bilateral orchiectomy). The trial design for KEYNOTE-991 is as follows: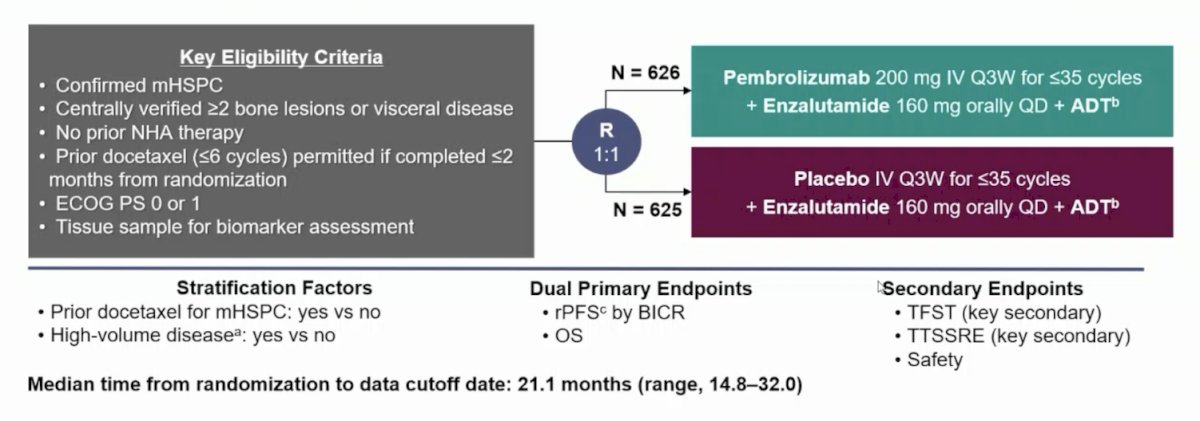
Dual primary endpoints were radiographic progression-free survival (rPFS) per PCWG-modified RECIST 1.1 by blinded independent central review and overall survival (OS). Key secondary endpoints included time to initiation of first subsequent anticancer therapy, time to first symptomatic skeletal-related event, and safety. From a statistical analysis standpoint, there was 16.6% power to detect a HR of 0.80 at an alpha of 2.5% (one-sided) for rPFS with 323 events. The overall type I error rate was strongly controlled at 2.5% (one-sided).
Between March 2, 2020, and August 9, 2021, 626 patients were randomized to pembrolizumab + enzalutamide and 625 to placebo + enzalutamide. As of October 31, 2022, the median follow-up time at first prespecified interim analysis was 21.1 months (range: 14.8−32.0). Baseline characteristics were generally balanced between arms and ∼10% of patients received prior docetaxel in each arm: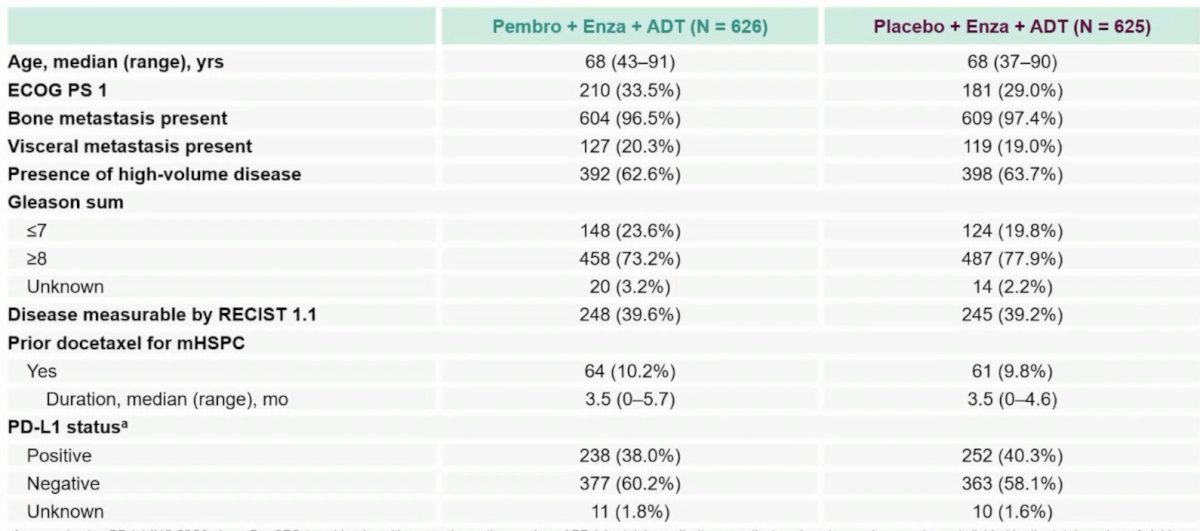
The primary endpoint of rPFS for pembrolizumab + enzalutamide vs placebo + enzalutamide was not met (median NR vs NR, HR 1.20, 95% CI 0.96−1.49):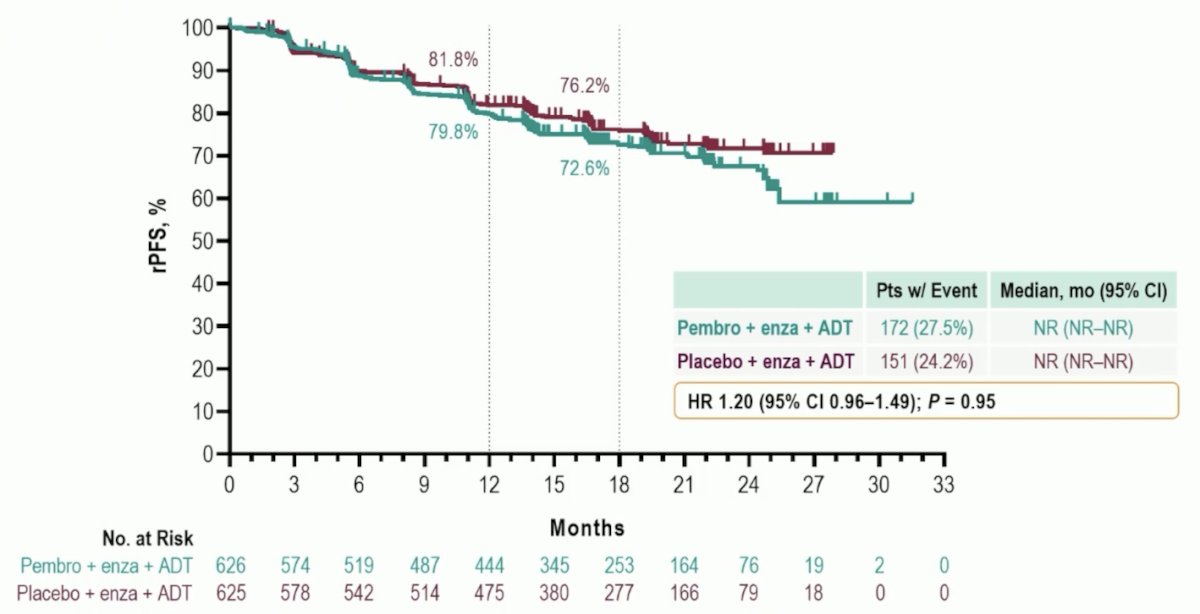
Based on the subgroup analyses, there were no groups that benefited from pembrolizumab + enzalutamide. In fact, those receiving docetaxel for mHSPC did worse with the combination therapy compared to placebo + enzalutamide (HR 2.37, 95% CI 1.19-4.73). The median OS was NR vs NR (HR 1.16, 95% CI 0.88−1.53):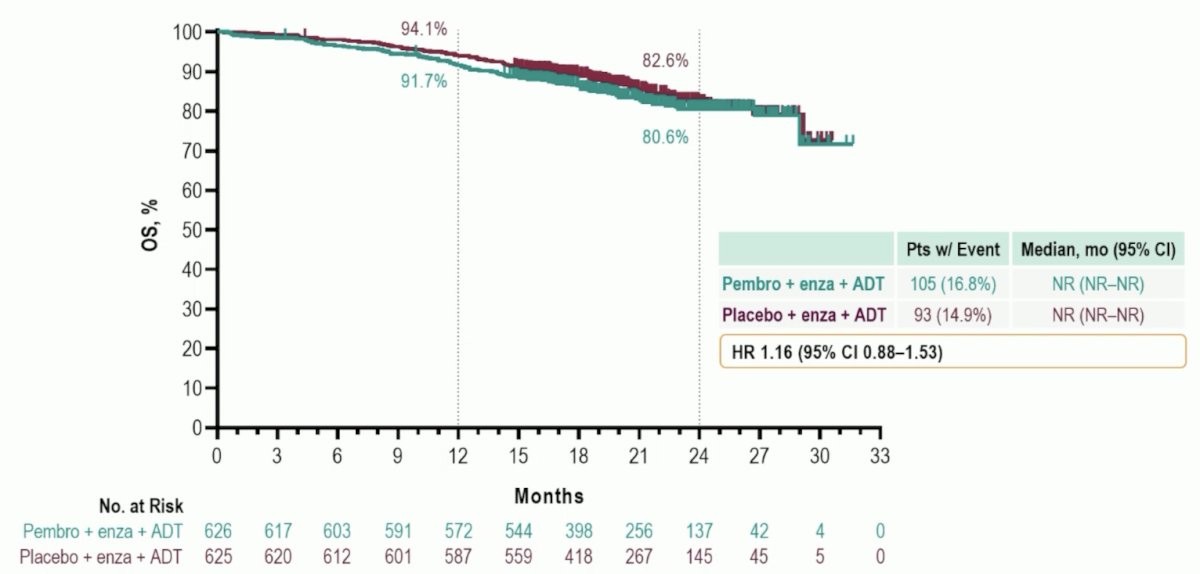
The median time to initiation of first subsequent anticancer therapy was NR in both arms (HR 1.24, 95% CI 1.01−1.54). Furthermore, median time to first symptomatic skeletal-related event was NR in both arms (HR 0.89, 95% CI 0.61−1.30). Serious adverse events occurred in 40.3% vs 23.2% of patients with pembrolizumab + enzalutamide vs placebo + enzalutamide. Any-grade and grade ≥3 treatment-related adverse events occurred in 88.0% vs 67.0% and 41.8% vs 13.9% of patients, respectively.
Dr. Gratzke concluded his presentation discussing results of the phase 3 KEYNOTE-991 trial assessing pembrolizumab + enzalutamide and ADT for patients with mHSPC with the following take-home points:
- The randomized phase 3 KEYNOTE-991 trial showed that the addition of pembrolizumab to enzalutamide and ADT did not significantly improve the primary endpoint of rPFS for patients with NHA-naïve mHSPC. The study was stopped for futility at the first prespecified interim analysis
- Secondary efficacy endpoints also did not appear to be improved with pembrolizumab + enzalutamide
- There was a higher frequency of any grade and grade >=3 treatment related adverse events and serious adverse events were observed in the pembrolizumab + enzalutamide group
- The role of immune checkpoint inhibition in prostate cancer remains unclear and further studies are needed to identify patients most likely to benefit
Presented by: Christian J. Gratzke, MD, Albert-Ludwigs-Universitat Freiburg, Freiburg, Germany
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Associate Professor of Urology, Georgia Cancer Center, Wellstar MCG Health, @zklaassen_md on Twitter during the 2023 European Society of Medical Oncology (ESMO) Annual Meeting, Madrid, Spain, Fri, Oct 20 – Tues, Oct 24, 2023.

