(UroToday.com) In the on-demand poster session of the European Society for Medical Oncology (ESMO) Annual Congress, Dr. Scott Tagawa reported results of a phase I/II study of dose-escalation study of a fractionated (dose-dense) treatment regime of 177Lu-PSMA-617 for metastatic castration-resistant prostate cancer (mCRPC). Previously reported, the initial phase I data from this study demonstrated no evidence of short-term dose-limiting toxicity.
In this study, men with progressive mCRPC following at least 1 AR-pathway inhibitor (ARPI) and taxane chemo (or ineligible/refuse) were enrolled. They were further required to have intact organ function and ECOG PS 0-2. Patients were treated with a single fractionated cycle of 177Lu-PSMA-617 on days 1 and 15 (7.4 to 22 GBq in dose-escalation cohort, followed by Simon 2-stage phase II study at 22GBq). While PSMA expression was not a prerequisite for treatment, baseline and post-treatment 68Ga-PSMA11 PET/CT and/or 177Lu-PSMA-617 SPECT were performed. The authors performed follow-up with serial PSA measurements, CT/bone scans, and circulating tumor cell (CTC; CellSearch) counts.
The authors included 50 men. In terms of baseline characteristics, the median PSA was 173.8 and 35 (70%) were deemed CALGB (Halabi) poor risk. In terms of disease burden, 47 (94%) had bone metastasis, 38 (76%) had LN metastasis, 12 (24%) had lung metastasis, and 11 (22%) had liver metastasis.
With respect to previous treatment, 29 (58%) had received ≥2 prior ARPI, 29 (58%) had received chemo, 14 (28%) had received radium-223, and 2 (4%) had received prior 177Lu-J591. 27 (54%) patients were treated at 22 GBq.
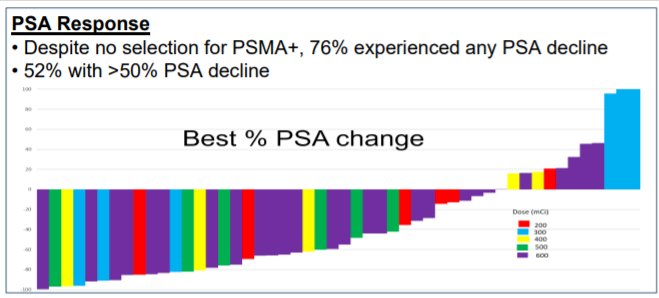
Of 50 included men, 40 (80%) experienced any PSA decline and 27 (54%) had >50% PSA decline. The median PFS was 5.6 months [95%CI 3.9-8.0], median radiographic PFS was 9.6 months [5.6-14.9], and median OS was15.2 months [10.8-27.0].
Of 31 with paired CTC counts who could be analysed, 16 (52%) had decreased CTC counts while 5 (16%) were stable and 10 (32.3%) converted to favorable/undetectable.
In terms of toxicity, adverse events included pain flare in 38 (76%), xerostomia in 27 (54%), nausea in 24 (48%), fatigue in 21 (42%), thrombocytopenia in 17 (34%), anemia in 10 (20%), transaminitis in 9 (18%), and neutropenia in 5 (10%). All AEs were Grade 1-2 except 4 (8%) patients with Grade 3 anemia.
On multivariable analysis, higher PSMA uptake (p<0.005) and dose (p=0.06) were associated with >50% PSA response while CALGB risk (p=0.032), prior chemo (0=0.041), higher PSMA uptake (0.085), and dose (p=0.072) were associated with OS.
The authors concluded that a single-cycle of fractionated-dose 177Lu-PSMA-617 is safe and may be efficacious. Additionally, patients were not pre-selected based on PSMA expression though most have post-treatment PSA decline with favorable biochemical and radiographic progression-free survival compared to historical non-PSMA controls and similar to PSMA-selected studies administering multiple cycles in a less dose-intense fashion.

