(UroToday.com) In this presentation, Dr. Toni Choueiri discussed patient reported outcomes (PROs) from KEYNOTE-564, a randomized phase 3 study of adjuvant Pembrolizumab versus placebo in patients with renal cell carcinoma (RCC). Results of the KEYNOTE-564 trial, initially presented at the ASCO 2021 Annual Meeting and recently published in the New England Journal of Medicine, demonstrated an improvement in disease-free survival at 24 months for patients randomized to receive adjuvant Pembrolizumab (77.3%) versus placebo (68.1%) (HR 0.68, 95% CI 0.53-0.87; P = 0.002).1 Overall survival data was immature and did not cross the pre-specified statistical threshold for this preliminary analysis, but at 24 month trended towards improvement for patients on the Pembrolizumab (96.6%) compared to the placebo (93.5%) arm (HR 0.54, 95% CI 0.30-0.96; P = 0.0164).
For the first time Dr. Choueiri discussed the patient reported outcomes for KEYNOTE-564. He first reviewed the adverse events (AEs) experienced on the trial. Grade 3-5 AEs occurred in 32.4% of patients who received pembrolizumab and in 17.7% of those who received placebo. Likewise, treatment-related Grade 3-5 serious all-cause AEs (20.5% versus 11.3%) and Grade3-5 treatment-related AEs (79.1% versus 53.4%) were higher on the Pembrolizumab than placebo arms. There were no treatment-related deaths on either arm.
The two PRO assessment instruments evaluated in this study were FKSI-DRS and EORTC QLQ-C30. FKS-DRS evaluates the most important symptoms associated with advanced kidney cancer. EORTC QLQ-C30 is a broader assessment that includes five functional scales, three symptom scales, and a global health status / quality of life scale. Based on prior literature, the threshold for a clinically meaningful change from baseline was 3 points for the FKSI-DRS and 10 points for EORTC QLQ-C30.
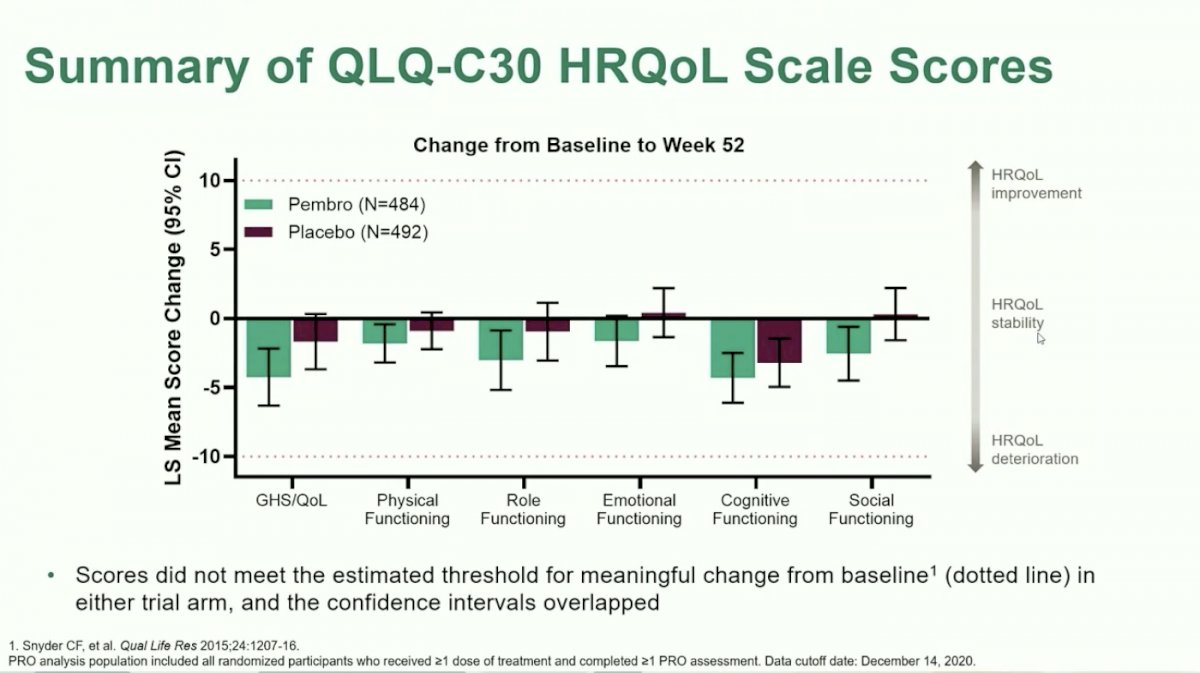
The first PRO discussed was change from baseline at 52 weeks, which was a pre-specified secondary endpoint. FKSI-DRS scores were minimally decreased on both arms, but did not meet the threshold for meaningful change in either trial arm. Further, the confidence intervals for the two arms overlapped, indicating no significant decrease in symptoms during the year of adjuvant treatment. Likewise, for the symptom scale scores and health-related quality of life (HRQoL) scores included in the EORTC QLQ-C30, there was no clinically meaningful change from baseline on either study arm at 52 weeks.


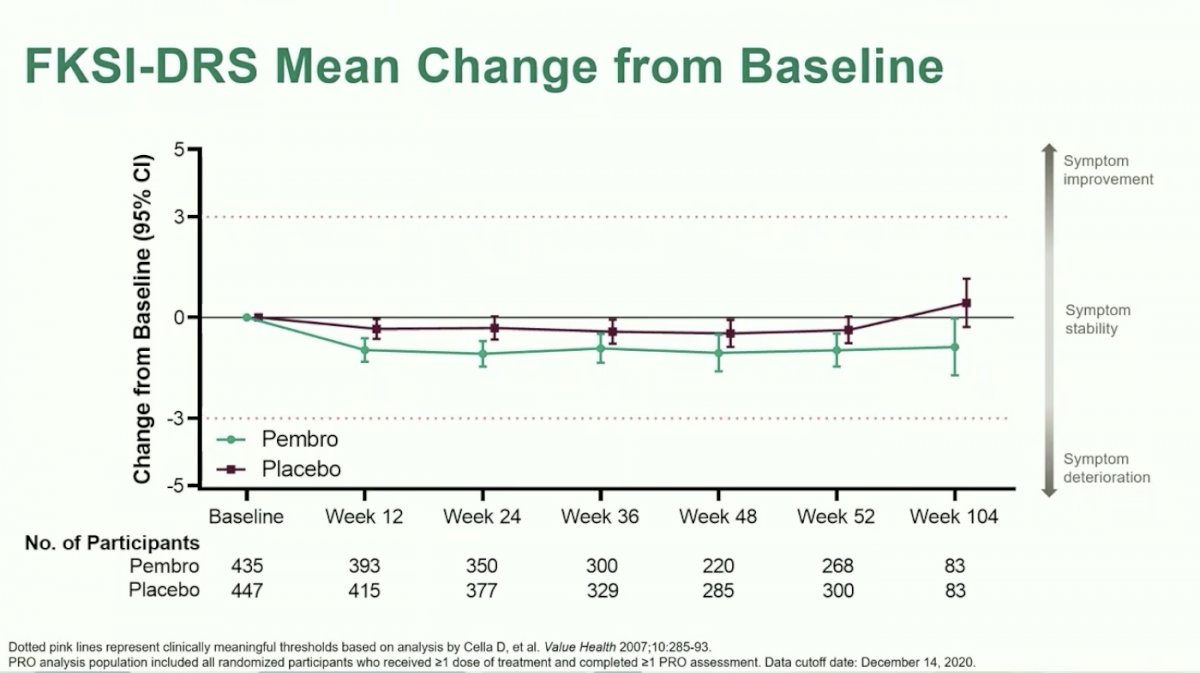
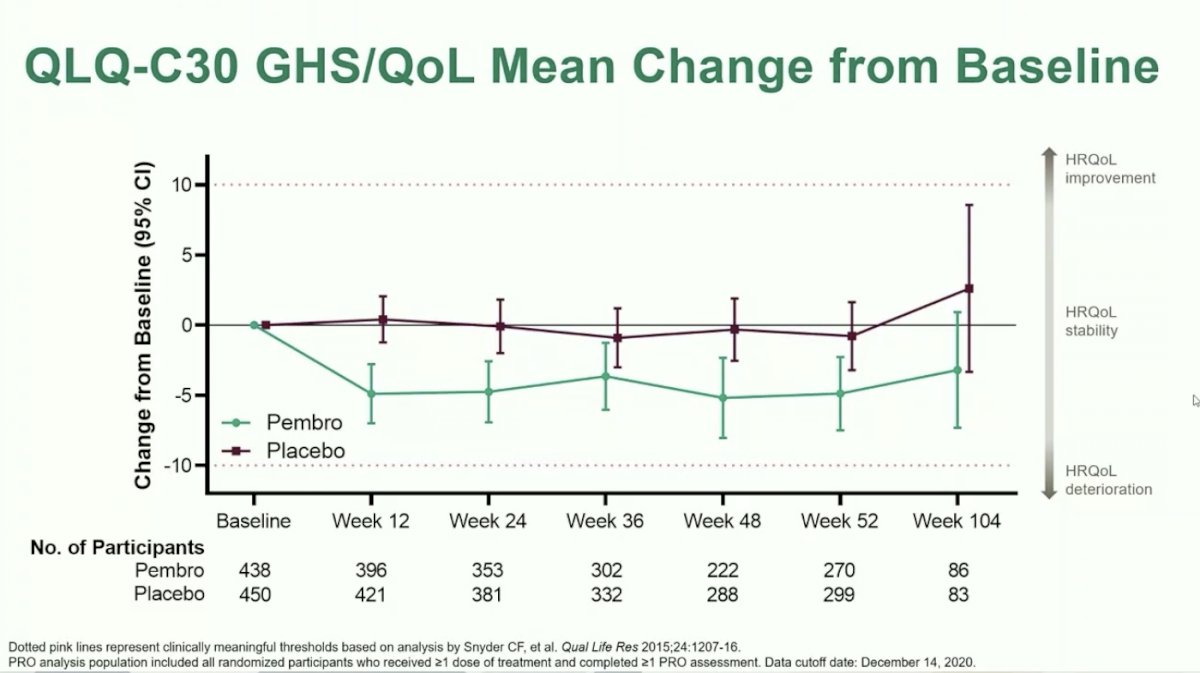
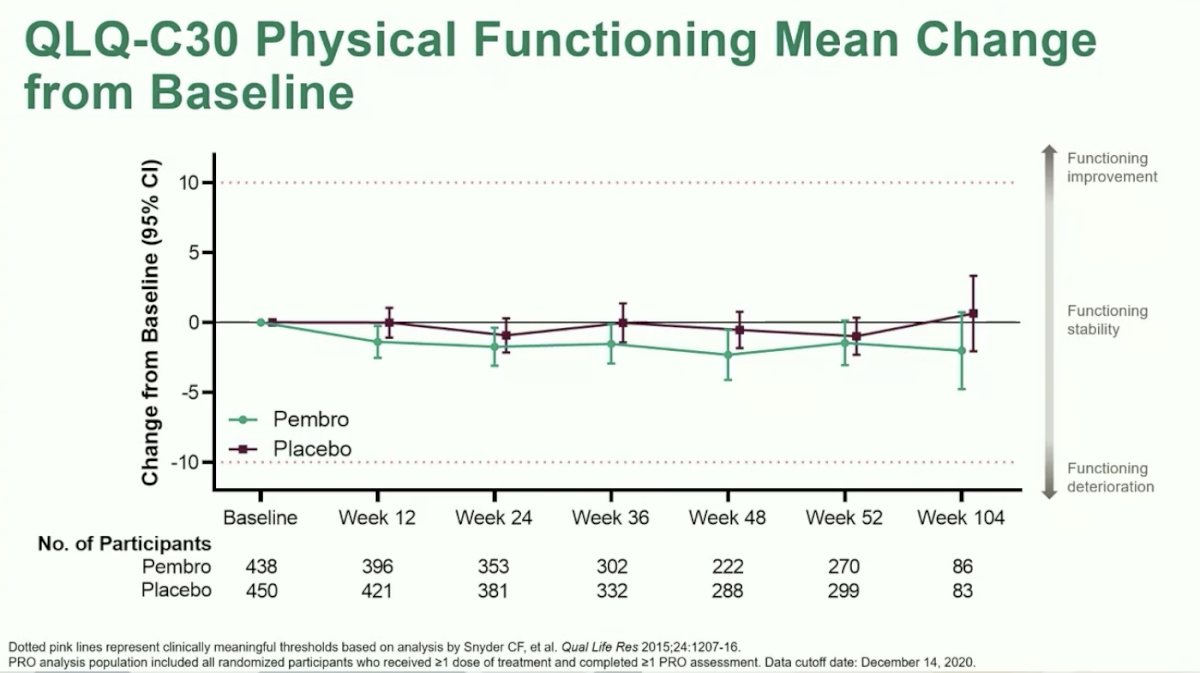
Dr. Choueiri moved on to discuss the first of two exploratory analyses: line plots for mean change from baseline over time. PRO scores were consistently numerically lower for patients treated with Pembrolizumab versus placebo over time, however, mean changes from baseline were not clinically significant. Likewise, there were no significant differences between arms.
`
The second exploratory analysis, overall summary of proportion of participants with improved/stable/deteriorated symptom score and HRQoL, likewise showed no clinically meaningful changes between the two arms. Across all PRO assessments, there was no difference in the proportion of patients treated with Pembrolizumab or placebo for the proportion of patients with improved versus stable versus deterioration from baseline.
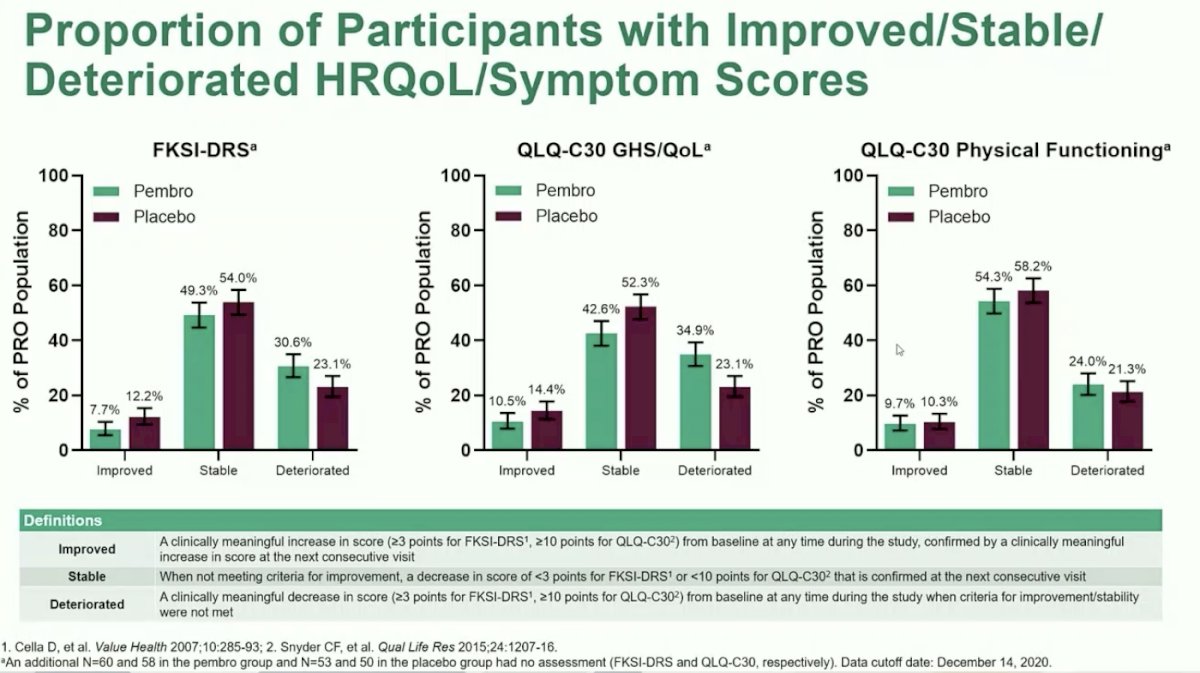
Dr. Choueiri concluded by emphasizing that in KEYNOTE-564, the first positive phase 3 study of adjuvant immunotherapy in RCC, there was no clinically meaningful difference in HRQoL or symptom scores for patients treated with Pembrolizumab compared to placebo. Scores on both treatment arms remained stable over the reported study period and disease-related symptoms were rarely severe at this disease stage. In summary, PROs from the KEYNOTE-564 study support the reported efficacy and safety findings from the first pre-specified analysis and establish Pembrolizumab as a potential new standard of care for patients with RCC in the adjuvant setting.
Presented by: Toni K. Choueiri, MD, Director, Professor of Medicine at Harvard Medical School, Medical Oncologist at Dana-Farber Cancer Institute, Director of the Lank Center for Genitourinary Oncology
Written by: Jacob Berchuck, MD, Genitourinary Medical Oncologist, Dana-Farber Cancer Institute (Twitter: @jberchuck) during the 2021 European Society for Medical Oncology (ESMO) Annual Congress 2021, Thursday, Sep 16, 2021 – Tuesday, Sep 21, 2021.
References:

