Presentation: Matthew Cooperberg, MD presented the role of genomic tests in active surveillance. First and foremost, he made it very clear – there are no prospective OR retrospective analyses of these genomic tests in the active surveillance setting. While studies are ongoing, the data has been extrapolated from actively treated patients.
There are a multitude of genomic tests/markers in the prostate cancer natural history paradigm. However, there are almost too many tests in each of the disease states. It becomes hard to manage them and the data behind them. His slide highlights the current status of these tests in the prostate cancer clinical timeline:
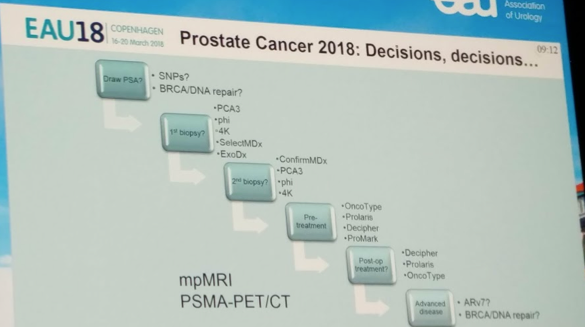
We are specifically looking at the pre-treatment phase. There are 3 primary genomic markers in this space: OncoType Genomic Prostate Score (Genomic Health), Decipher genomic classifier (GenomeDx), and Prolaris cell cycle score (Myriad). All three have been shown in numerous studies to improve multivariable clinical models to predict post-treatment endpoints – but studies in AS are ongoing.
He began by highlighting the key principles of biomarkers – a putative biomarker must improve on an existing, multivariable clinical model, ideally a previously validated one. In prostate cancer, there are established nomograms and validated models already in use that are already pretty good at predicting outcomes, including the UCSF based CAPRA / CAPRA-S scores (C-index 0.79). Hence, the bar is high for improved accuracy.
The specific questions that need to be addressed in the AS setting:
1) What is the optimal clinical base model?
2) What is the appropriate endpoint? Unfortunately, the best would be PCSM/OM – but in men with low-risk disease, this may take decades to address.
- Surrogates have been developed: adverse pathology at time of surgery, recurrence after treatment, development of metastases
- But, the key question is, when does the window for cure close?
He made a very important statement: choosing the wrong surrogate endpoint may doom a biomarker to failure, even if it has clinical import.
He briefly touched on urine tests, such as PCA3 and TMPRSS2:ERG, but unfortunately both of these were not associated with higher risk disease independent of clinical variables. Hence, they did not improve upon multivariable models.
Ultimately, while we would love to have “pregnancy” test for active surveillance (ie: yes / no to treatment), all of these tests merely shift the shade of grey towards AS or towards treatment.
His quick jab at MRI in prostate cancer AS management is that recent studies that have finally started to look at interobserver reliability have been quite concerning. Even at high volume MR centers, even a PIRADS 5 lesion may be highly variable in terms of predicting actionable disease. He cited the recent Stanford paper (Sonn GA EU 2017).
The only guidelines that address this issue is the 2016 ASCO endorsement of the CCO guidelines – AS protocols may include ancillary tests that are still under investigation. These can be mpMRI and/or genomic tests. These are indicated when patient’s clinical history is discordant with pathology. mpMRI should not be used as a biopsy substitute.
Conclusions:
1. Existing tissue-based biomarkers have independent prognostic value – but not yet in AS series
2. Markers and imaging may help in decision-making, particularly in borderline patients (high-volume Gleason 6 or low volume Gleason 7)
3. Genomics will help tailor AS – if not now, then very soon
Presented by: Matthew Cooperberg, MD San Francisco, CA UCSF
Read More:
Part 2 of Debate, Presented by: Professor Olivier Rouvière, MD, PhD from Lyon, France: Clinical Data and Imaging are Doing the Job
Written by: Thenappan Chandrasekar, MD Clinical Fellow, University of Toronto, twitter: @tchandra_uromd at the 2018 European Association of Urology Meeting EAU18, 16-20 March, 2018 Copenhagen, Denmark

