In 2018, we have learned that Gleason 3 resembles normal cells in most cases (90%) and harbor a metastatic potential of 0. The only important significance of high volume Gleason grade group 1 is the increased risk of coexistent higher-grade cancer. It is important to be aware of two critical concepts:
1. Misattribution of the grade in the biopsy, which is quite common and occurs in 25-30% of cases.
2. Biological grade progression that occurs over time (1-2% per year)1
There are almost no well-documented cases of pathologically proven Gleason 6 cancers that have metastasized. In a study assessing 12000 Gleason 6 cancers treated with radical prostatectomy with 20-year follow-up – the prostate cancer-specific mortality was 0.2% at 20 years 2 (and a second review of these cases showed a higher-grade disease). There is also data showing that genomic alteration is quantitively and not qualitatively different between different prostate cancer grades.3 Data has also been published to show that there is a diverse genomic landscape of clinically low-risk prostate cancer. This data showed that Gleason Grade group 1, and 2 had 2% and 14% score, respectively, of genetic aberrancy and aggressivity.4
In the Toronto Surveillance cohort, there are 1400 patients with a median follow-up of 8.9 years (0.5-19.8 years). For these patients, serial PSA was obtained every six months and biopsy was performed yearly at the beginning, and then every 3-5 years. MRI was introduced only in 2012. The cohort consists of 78% low-risk patients, and 22% intermediate risk patients. A total of 30 patients from the entire cohort developed metastases with 15 of them dying of prostate cancer, four dying of other causes, and 11 currently alive with metastases.
The selection criteria for active surveillance among the various centers who have published their data is quite different, as shown in figure 1. This has obviously had an impact on the results of these cohorts as can be seen in figure 2. When assessing the long-term outcomes between the Toronto cohort and the Johns Hopkins cohort (Figure 3), significant differences can be seen due to the much more liberal criteria for surveillance being used by the Toronto group (prostate cancer mortality of 0.5% in Johns Hopkins vs. 5% in the Toronto cohort – mostly due to the inclusion of Gleason 7 patients).
Figure 1- Different Active Surveillance Protocols:
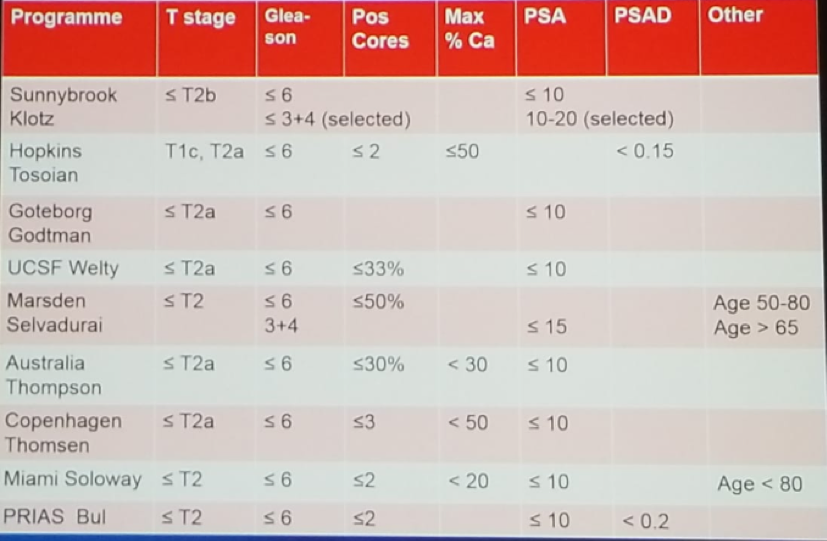
Figure 2 - Active Surveillance Outcomes:

Figure 3 – Long Term Outcomes of Active Surveillance:

Active surveillance has been shown to be safe in young patients as well (<60) with no difference in treatment rates between men younger than 60 and men older than 60 (74% vs. 71%), and with similar metastasis-free survival (99.7% vs. 99%), and cancer-specific survival (100% vs. 99.7%). 5 The only important limitation to this study is the lack of the required 30 years of follow-up.
According to Dr. Klotz, men with intermediate-risk disease who can be managed with active surveillance include:
1.Those with Gleason 3 + <=5% pattern 4 (often artifactually upgraded).
2.Low volume Gleason grade group 2 with negative mpMRI
3.Low volume Gleason grade group 2 with favorable tissue-based biomarker scores. However, we need the right test in the right patient with a risk in the “sweet spot.”
4.The balance between risk, age, and co-morbidity.
Before concluding his talk, Dr. Klotz discussed the topic of active surveillance in patients with genetic mutations in ATM and BRCA 1 and 2. These have been associated with grade reclassification, with an impressive hazards ratio of 2.7 for men with BRCA2.6 This raises the question of whether these men should even be offered active surveillance.
In conclusion, active surveillance is a robust strategy for many cancers with an indolent phenotype. It manifests an opportunity to reduce morbidity, cost, and enhance the appeal of early detection. However, it requires patient and physician (and payer) buy-in. It needs to be congruent with the emerging era of molecular medicine, and it presents an opportunity for concurrent health maintenance interventions.
Presented by: Laurence Klotz, MD, FRCSC, Professor, Department of Surgery, University of Toronto, Chief, Division of Urology, Sunnybrook Health Sciences Center, Chair, Canadian Uro-Oncology Group, and NCIC GU Site Group, Editor in Chief (founding), Canadian Journal of Urology, Chair, Global GU Oncology Group
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, Twitter: @GoldbergHanan at the CUOS – Canadian Uro-Oncology Summit 2019, #CUOS19 January 10-12, 2019 Westin Harbour Castle, Toronto, Ontario, Canada
References:
1. Inoue LY et al. Stat Med 2014
2. Eggener S, J Urol 2011
3. Rubin M. et al. Eur Urol 2016
4. Cooperberg M. et al. Eur Urol 2018
5. Klotz L. et al. AUA 2018
6. Carter HB. et al. Eur Urol 2018

