The cancer immune cycle is as follows:
- The release of cancer cell antigens at the time of death
- Cancer antigen presentation by dendritic cells and antigen-presenting cells
- Priming and activation of T-cells by antigen-presenting cells
- Trafficking of T-cells to tumors
- Infiltration of T-cells into tumors
- Recognition of cancer cells by T-cells
- The killing of cancer cells
CTLA-4 is an immune checkpoint receptor on T-cells that plays a key role in preventing T-cell overactivation. Tumor cells use the CTLA-4 pathway to suppress the initiation of an immune response, resulting in decreased –cell activation and ability to proliferate into memory cells. CTLA-4 signaling diminishes the ability of memory T-cells to sustain a response, damaging a key element of durable immunity. PD-1 is an immune checkpoint receptor on cytotoxic T-cells that plays a key role in T-cell exhaustion and prevention of autoimmunity. Tumor-infiltrating T-cells across solid tumors and hematologic malignancies display evidence of exhaustion, including upregulation of PD-1. PD-1 blockade reinvigorates exhausted T-cells and restores their cytotoxic immune function.
Lori Wood, MD, then presenting on the role of immune checkpoint inhibitors in metastatic urothelial carcinoma. We know that second line chemotherapy demonstrates a response rate of 10-20%, with a time to progression of 2-4 months, and a median overall survival of 5-9 months. The approved immune checkpoint inhibitors for metastatic urothelial carcinoma are as follows:
- PD-1 (on T-cells):
- Nivolumab
- Pembrolizumab
- PD-L1 (on the cancer cell):
- Atezolizumab
- Durvalumab
- Avelumab
Initial studies included multiple phase I/II studies, leading to FDA approval primarily based on phase II studies, with similar approval in Canada. As follows is a chart of immune checkpoint inhibitors in metastatic urothelial carcinoma:
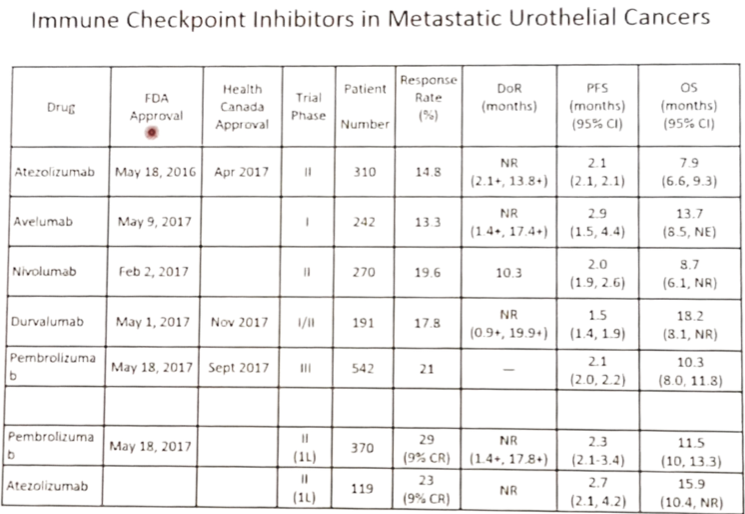
Several important second-line phase III trials in metastatic urothelial carcinoma have recently reported initial results. These patients were those that progressed after platinum-based chemotherapy in the metastatic setting or those that had a recurrence within one year of adjuvant/neoadjuvant chemotherapy. The phase 3 KEYNOTE-045 study comparing pembrolizumab and investigator’s choice of chemotherapy (paclitaxel, docetaxel, or vinflunine) reported results after the second planned interim analysis (at which point the trial was stopped)1. The study found a median OS of 10.3 months (95%CI 8.0-11.8) in the pembrolizumab group, compared with 7.4 months (95%CI 6.1-8.3) in the chemotherapy group (HR 0.73, 95%CI 0.59-0.91). Furthermore, the median OS among patients who had a tumor PD-L1 combined positive score (CPS) of ≥10% was 8.0 months (95%CI 5.0-12.3) in the pembrolizumab group, as compared with 5.2 months (95%CI 4.0-7.4) in the chemotherapy group (HR 0.57, 95%CI 0.37-0.88). Based on these results, pembrolizumab was FDA approved for the treatment of locally advanced or metastatic urothelial carcinoma in the second line. The phase III IMvigor211 study tested the efficacy of atezolizumab versus chemotherapy among patients progressing on platinum-based chemotherapy [2]. There were 931 patients randomized to receive atezolizumab (n=467) or chemotherapy (n=464). In the IC2/3 population (n=234), overall survival did not differ significantly between patients in the atezolizumab group and those in the chemotherapy group (median 11.1 months, 95%CI 8.6-15.5 vs 10.6 months 95%CI 8.4-12.2) (HR 0.87, 95%CI 0.63-1.21). An exploratory analysis of the intention-to-treat population showed durable responses in line with previous phase II data from IMvigor 210 for atezolizumab in this setting. Unfortunately, atezolizumab was not associated with significantly longer OS than chemotherapy in patients overexpressing PD-L1 (IC2/3). Atezolizumab was well tolerated compared to chemotherapy, with less all grade (60.9% vs 90.2%) and grade 3-5 (15.0% vs. 49.4%) treatment-related adverse events. Furthermore, treatment discontinuation rates were less with atezolizumab (5.6% vs 11.0%).
In summary, Wood noted:
- There are many PD-1 pathway immune checkpoint inhibitors and many new inhibitors being studied
- For metastatic disease, in Canada, these agents are only approved in the second line and not approved for cisplatin ineligible at this point in time
- There are only two phase III trials currently, and only one with positive results
- Indeed, there are profound results in some patients
- Who are the metastatic patients with profound responses?
- Should we use in the first line setting? Alone or in combination with chemotherapy?
- Should we be using in the neoadjuvant or adjuvant setting?
- Should we be using in NMIBC?
- Any high-grade recurrence after induction BCG + first round of maintenance BCG, or two rounds of induction BCG [Ta/CIS failure to achieve a complete response at 6 months]
- An exception is high-grade T1 disease at three months (after induction BCG only), which is considered “unresponsive” [T1 failure to achieve a complete response at 3 months]
- For patients who achieve a complete response on induction/maintenance BCG: any high-grade recurrence within 6 months of last dose of BCG [relapse of high-grade recurrence within 6 months of the last dose of BCG after a prior complete response]
Black then highlighted several common clinical caveats:
Recurrent low-grade Ta NMIBC does NOT constitute BCG-unresponsive NMIBC in this context Do NOT deem BCG treatment to have failed after induction BCG only in patients with Ta and CIS >Ensure prostatic urethra and upper tracts are clear in BCG-unresponsive patients, considering that in these sanctuary sites there is up to 50% involvement
Data from the SWOG S0353 study for BCG-unresponsive NMIBC demonstrates that the best we can do with intravesical maintenance gemcitabine for BCG “refractory” NMIBC is a 28% recurrence-free survival (RFS) at 1-year, and 20% RFS at 2-years [3]. Based on these results, according to Black “our best intravesical salvage therapy is not good enough.” The guidelines state that in these situations, the treatment is radical cystectomy, whereas many of the experts state that it may be reasonable to administer one more round of intravesical therapy before proceeding to cystectomy for high-grade Ta and CIS (but always radical cystectomy for high-grade T1).
The rationale for testing immune checkpoint inhibitors in NMIBC is (i) there is the efficacy of immunotherapy in NMIBC (BCG), (ii) there is an expression of PD-L1 in Ta, T1 and CIS in patients previously treated with BCG, and (iii) there is pre-clinical efficacy data from syngeneic mouse models. There are currently two ongoing immunotherapy trials ongoing in the BCG unresponsive disease state. The SWOG S1605 trial:

And the KEYNOTE-057 trial:

Black notes that with regards to clinical trials in NMIBC, there are many trials to be done and this requires urologists to “engage”. This will require more collaboration with medical oncology and strict eligibility criteria are unfortunate but essential in order to make the results interpretable.
Black concluded with discussing two phase II neoadjuvant trials that recently presented initial results last month at ASCO. The PURE-01 trial is an open-label, single-arm, phase 2 study evaluating pembrolizumab prior to radical cystectomy4. The primary outcome was a pathologic complete response (pT0) at the time of radical cystectomy in the intention to treat (ITT) population. The first stage of enrollment included 43 patients, including 35 males/7 females, with 37.2% of patients with cT2N0 disease, 58.1% with cT3N0, and 4.7% of patients with T2-3N1. At the time of this analysis, there were 17/43 patients that were pT0 (39.5%, 95%CI: 26.3-54.4) and 5 <pT2 (total <pT2 rate: 51.2%). The ABACUS trial is a single arm, phase II study investigating two cycles of atezolizumab (1200mg every three weeks) prior to radical cystectomy among patients with T2-4N0M0 urothelial carcinoma5. Among 68 patients, the median age was 71 years (range 53-85), and the baseline pT2 rate was 71%, pT3 was 22%, and pT4 was 7%. The pathologic complete response rates were as follows: (i) all patients: 20/68 (29%) – pT0, n=16; pTis, n=4; (ii) PD-L1 positive patients: 10/25 (40%); (iii) PD-L1 negative patients: 5/31 (16%); (iv) cT2 patients: 17/48 (35%); (v) cT3-T4 patients: 3/20 (15%).
Black concluded with several summary points:
- BCG-unresponsive NMIBC is an important concept for clinical trials and routine practice
- Immune checkpoint blockade will likely be a part of routine therapy for NMIBC and localized MIBC in the near future single-agent trials are just the tip of the iceberg
- Urologists need to familiarize themselves with these agents and also support ongoing clinical trials.
References:
1. Bellmunt J, de Wit R, Vaughn DJ, et al. Pembrolizumab as Second-Line Therapy for Advanced Urothelial Carcinoma. N Engl J Med 2017;376(11):1015-1026.
2. Powles T, Duran I, van der Heijden MS, et al. Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): A multicentre, open-label, phase 3 randomized controlled trial. Lancet 2018;391:748-757.
3. Skinner EC, Goldman B, Sakr WA, et al. SWOG S0353: Phase II trial of intravesical gemcitabine in patients with nonmuscle invasive bladder cancer and recurrence after 2 prior courses of intravesical bacillus Calmette-Guerin. J Urol 2013;190(4):1200-1204.
4. Necchi A, Briganti A, Bianchi M, et al. Preoperative pembrolizumab before radical cystectomy for muscle-invasive urothelial bladder carcinoma: Interim clinical and biomarker findings from the phase 2 PURE-01 study. ASCO 2018 abstr 4507.
5. Powles T, Rodriguez-Vida A, Duran I, et al. A phase II study investigating the safety and efficacy of neoadjuvant atezolizumab in muscle-invasive bladder cancer (ABACUS). ASCO 2018 abstr 4506.
Moderator: Bobby Shayegan, MD, McMaster University, Hamilton, Ontario, Canada
Speaker: Lori Wood, MD, Dalhousie University, Halifax, Nova Scotia, Canada; Peter Black, MD, University of British Columbia, Vancouver, British Columbia, Canada
Written By: Zachary Klaassen, MD, Urologic Oncology Fellow, University of Toronto, Princess Margaret Cancer Centre Twitter: @zklaassen_md at the 73rd Canadian Urological Association Annual Meeting - June 23 - 26, 2018 - Halifax, Nova Scotia

