(UroToday.com) The Society of Urologic Oncology session at the 2021 Virtual AUA annual meeting’s bladder cancer session included a presentation by Dr. Peter Black discussing what is new for biomarkers in bladder cancer. Dr. Black highlighted that there are different types of biomarkers based on the disease state of interest:
- Non-muscle invasive bladder cancer (NMIBC): urine markers for detection and monitoring
- Muscle invasive bladder cancer: (i) markers to predict response to neoadjuvant chemotherapy, and (ii) ctDNA to guide adjuvant therapy after radical cystectomy
- Metastatic urothelial carcinoma: markers for guiding systemic/targeted therapy
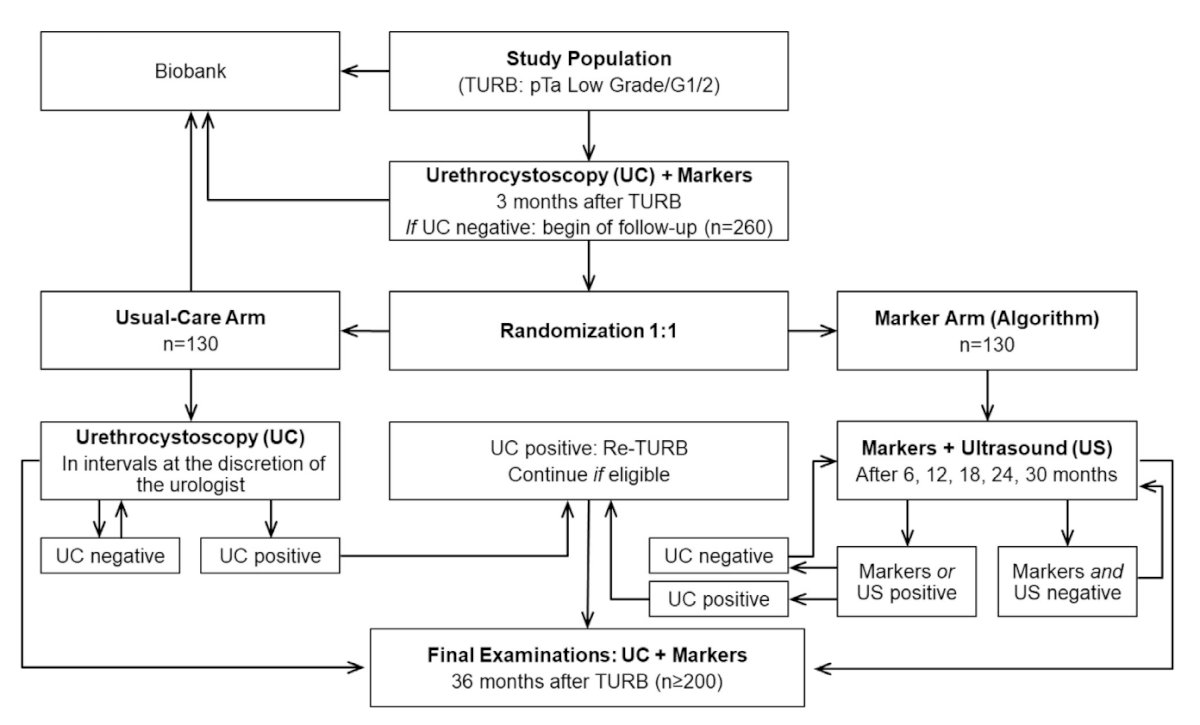
Dr. Black notes that there is an extensive history of biomarkers in bladder cancer dating back to 1970 with the advent of urine cytology, however what we are still missing is intervention trials comparing standard of care to marker-directed management.1 One such study is the UroFollow trial that has been designed as a prospective randomized study comparing a noninvasive marker-based (UroVysion, NMP22, urine cytology, and ultrasound) follow-up with the current standard of care over a period of 3 years. The following figure is the UroFollow study design:
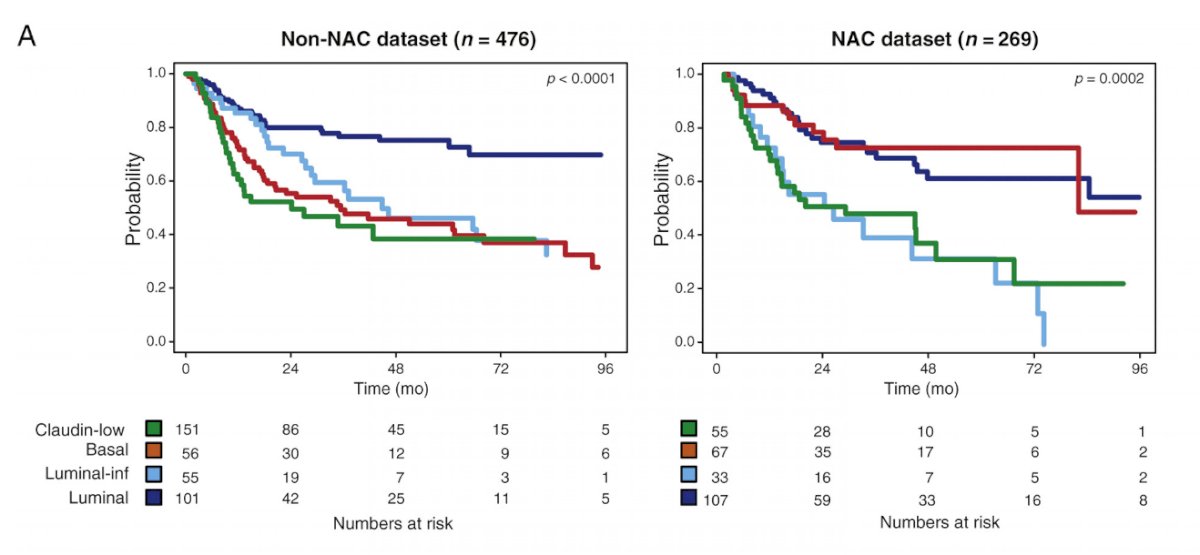
In the neoadjuvant chemotherapy disease space, there are several molecular predictors of response: (i) molecular subtypes – basal/squamous, luminal papillary, etc; (ii) COXEN model – RNA expression-based molecular marker; and (iii) DNA damage repair gene alterations – ERCC2, ATM, etc. To investigate the ability of molecular subtypes to predict pathological downstaging and survival after neoadjuvant chemotherapy, Seiler et al.3 assessed pre-neoadjuvant chemotherapy transurethral specimens from 343 patients with muscle-invasive bladder cancer. Overall survival (OS) according to subtype was analyzed and compared with OS in 476 non-neoadjuvant chemotherapy cases (published datasets). The genomic subtyping classifier was able to predict four consensus molecular subtypes with high accuracy (73%), and clinical significance of the predicted consensus subtypes could be validated in independent neoadjuvant chemotherapy and non-neoadjuvant chemotherapy datasets. Luminal tumors had the best OS with and without neoadjuvant chemotherapy, whereas claudin-low tumors were associated with poor OS irrespective of treatment regimen. Basal tumors showed the most improvement in OS with neoadjuvant chemotherapy compared with surgery alone.
The SWOG S1314 COXEN clinical trial was a biomarker validation trial with the following trial design:4
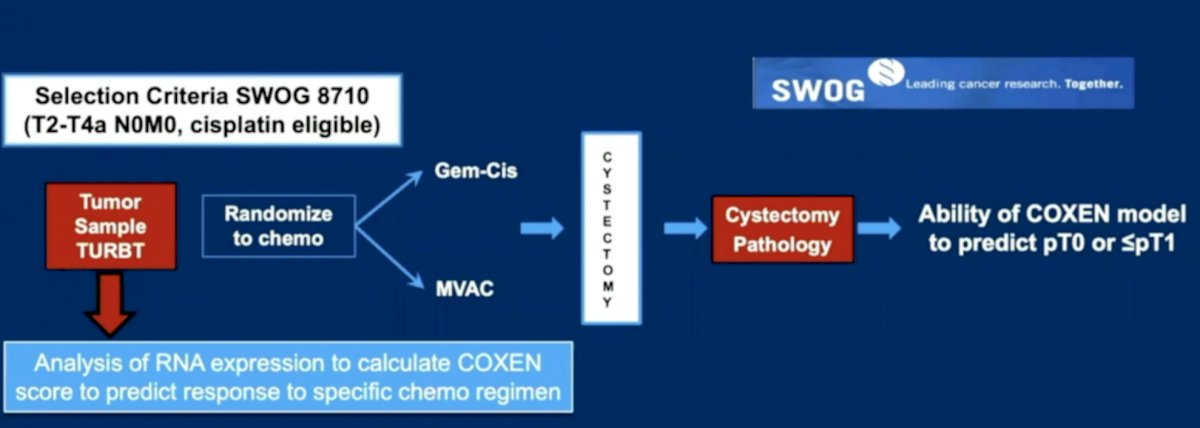
Among 167 evaluable patients, the odds ratio (OR) for pT0 with the gemcitabine-cisplatin gene expression model score in gemcitabine-cisplatin treated patients was 2.63 (95% CI 0.82-8.36]; for the ddMVAC COXEN gene expression model score with ddMVAC treatment, the OR was 1.12 (95% CI 0.42-2.95). The gemcitabine-cisplatin gene expression model score was applied to pooled arms (gemcitabine-cisplatin and ddMVAC) for downstaging with an OR of 2.33 (95% CI, 1.11-4.89).
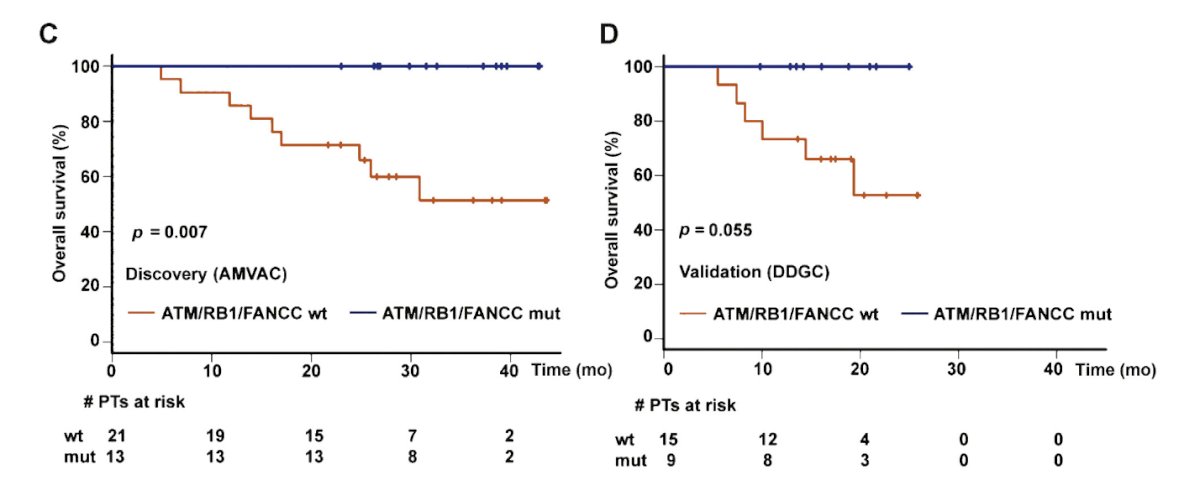
Mutations in DNA damage repair genes also predict survival after neoadjuvant chemotherapy. Work from Dr. Betsy Plimack published in European Urology in 20155 showed that patients with a pathologic complete response had more alterations than those with residual tumor in both the discovery (p=0.024) and validation (p=0.018) sets. In the discovery set, alteration in one or more of the three DNA repair genes ATM, RB1, and FANCC predicted pathologic response (p<0.001; 87% sensitivity, 100% specificity) and better overall survival (p=0.007).
The RETAIN trial initially presented results at the GU ASCO 2021 meeting. This is an adaptive trial with the following flow diagram:

To date, 71 patients have been enrolled in the trial, with 38 having no identifiable mutation, and 33 patients with a mutation. There were 25 T0 patients on re-TURBT after neoadjuvant chemotherapy (76%) with these patients staring on surveillance; 65% ultimately had a recurrence and the primary outcomes of 2-year metastasis free survival is still pending.
In the adjuvant setting, circulating tumor DNA is prognostic and predictive after adjuvant atezolizumab.6 Among 581 patients, ctDNA testing at the start of therapy (cycle 1 day 1) identified 214 (37%) patients who were positive for ctDNA and who had poor prognosis (observation arm HR 6.3, 95% CI 4.45-8.92). Notably, patients who were positive for ctDNA had improved disease-free survival and OS in the atezolizumab arm versus the observation arm (disease-free survival HR 0.58, 95% CI 0.43-0.79, OS HR 0.59, 95% CI 0.41-0.86).
In advanced bladder cancer, Dr. Black emphasizes that the treatment landscape has rapidly evolved over the last several years. Based on the change in the label in September 2021 from the FDA to pembrolizumab or atezolizumab in first-line metastatic urothelial carcinoma, regardless of PD-L1 status, there is currently no role of PD-L1 immunohistochemistry in any disease state:
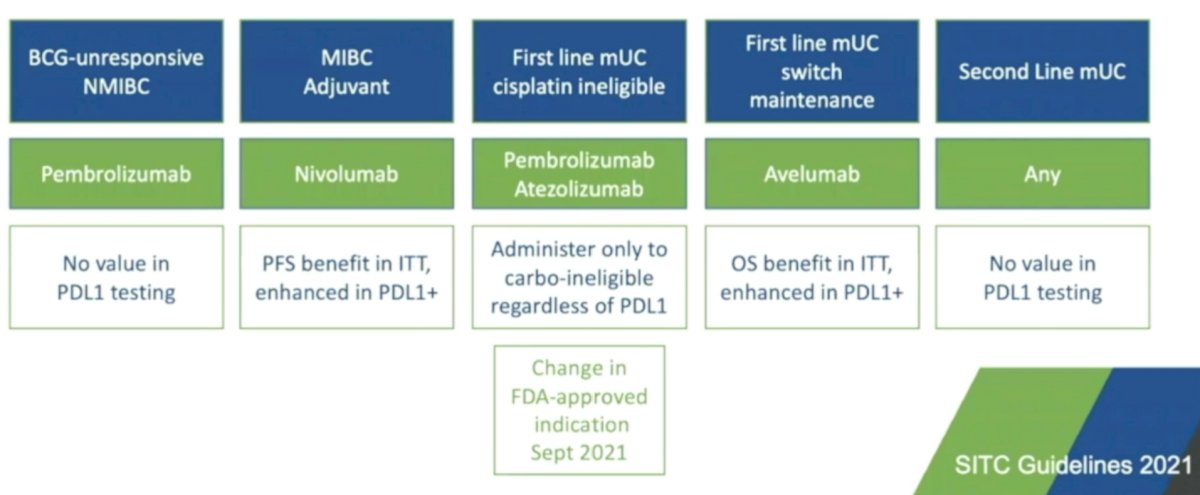
Finally, erdafitinib is approved in platinum refractory metastatic urothelial carcinoma among patients screened positive for FGFR translocations or mutations.
Dr. Black concluded his presentation with the following take home messages for what is new in biomarkers:
- The quest for the optimal urine marker continues
- Markers to predict response to neoadjuvant chemotherapy (especially DNA damage repair gene mutations and molecular subtypes) is being tested in a randomized clinical trial
- ctDNA may be prognostic and predictive of response to adjuvant immune checkpoint blockade, with prospective validation pending
- PD-L1 immunohistochemistry currently has no role in guiding immunotherapy in urothelial carcinoma
- FGFR testing is essential to guide anti-FGFR therapy in metastatic urothelial carcinoma
Presented by: Peter C. Black, MD, FACS, FRCSC, Khosrowshahi Family Chair, Professor Department of Urologic Sciences, Vancouver Prostate Cancer, University of British Columbia, Vancouver, BC, Canada
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2021 American Urological Association, (AUA) Annual Meeting, Fri, Sep 10, 2021 – Mon, Sep 13, 2021.
References:
- Lotan Y, Black PC, Caba L, et al. Optimal trial design for studying urinary markers in Bladder Cancer: A collaborative review. Eur Urol Oncol. 2018 Aug;1(3):223-230.
- Benderska-Soder N, Hovanec, Pesch B, et al. Toward noninvasive follow-up of low-risk bladder cancer – Rationale and concept of the UroFollow trial. Urol Oncol. 2020 Dec;38(12):886-895.
- Seiler R, Al Deen Ashab H, Erho N, et al. Impact of molecular subtypes in muscle-invasive bladder cancer on predicting response and survival after neoadjuvant chemotherapy. Eur Urol. 2017 Oct;72(4):544-554.
- Flaig TW, Tangen CM, Daneshmand S, et al. A randomized phase II study of Coexpression Extrapolation (COXEN) with Neoadjuvant Chemotherapy for Bladder Cancer (SWOG S1314; NCT02177695). Clin Caner Res. 2021 May 1;27(9):2435-2441.
- Plimack ER, Dunbrack RL, Brennan TA, et al. Defects in DNA repair genes predict response to neoadjuvant cisplatin-based chemotherapy in muscle-invasive bladder cancer. Eur Urol. 2015 Dec;68(6):959-967.
- Powles T, Assaf ZJ, Davarpanah N, et al. ctDNA guiding adjuvant immunotherapy in urothelial carcinoma. Nature. 2021 Jl;595(7867):432-437.

