(UroToday.com) The 2023 American Society of Clinical Oncology Genitourinary (ASCO GU) cancers symposium held in San Francisco, CA between February 16th and 18th was host to a prostate cancer trials in progress poster session. Dr. Daniel Spratt presented the study design for OPTYX, a multi-center, prospective observational study of patients with prostate cancer being treated with relugolix (ORGOVYX®).
Androgen deprivation therapy, in the form of gonadotropin-releasing hormone (GnRH) receptor agonists or antagonists, is the backbone treatment for patients with advanced prostate cancer. Relugolix (ORGOVYX®) is an oral non-peptide GnRH receptor antagonist approved for patients with advanced prostate cancer, following results of the HERO trial. The HERO trial was a non-inferiority phase III trial of 622 patients randomly assigned 2:1 to either relugolix 120 mg orally once daily or leuprolide injections every 3 months for 48 weeks. Relugolix was shown to maintain castration levels (<50 ng/dL) through 48 weeks in 96.7% of patients, compared to 88.8% in those receiving leuprolide. Relugolix was furthermore shown to be associated with a more favorable cardiovascular risk profile. Among all study patients, the incidence of major adverse cardiovascular events was 2.9% in the relugolix group, compared to 6.2% in the leuprolide group (hazard ratio, 0.46; 95% CI, 0.24 to 0.88).1 The importance of real world registries is to demonstrate how treatment decisions in everyday clinical practice affect clinical outcomes, quality of life, and total cost of care. This has been demonstrated in other registries, including CaPSURE, AQUA, and IRONMAN. The goal of OPTYX was to evaluate the safet and effectiveness of relugolix for the treatment of patients with advanced prostate cancer in a real-world setting.
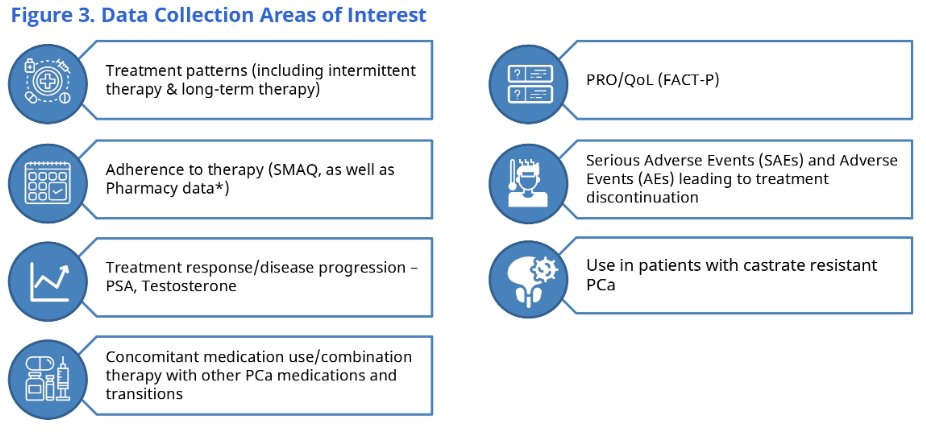
This registry will prospectively enroll patients ages 18 years or older with prostate cancer and planned total treatment with relugolix for at least 4 months. Patients who have planned initiation with relugolix within one month prior to the time of study enrollment will be included. This study will prospectively evaluate the real-world experiences of patients treated with relugolix, through data collection on patients selected for treatment, treatment patterns, adherence, patient reported quality of life, and safety data. Enrollment sites will include academic and university-based centers, independent practices, community hospitals, and integrated care systems (such as Veterans Affairs [VA]).


Important data following cessation of relugolix will also be obtained, in the form of observation of the disease clinical course, health outcomes (including comorbidities), and health-related quality-of-life. This registry will plan to enroll 1000 patients, with a follow-up of five years. Patient reported outcomes will be assessed using the Functional Assessment of Cancer Therapy – Prostate (FACT-P), which will assess health-related quality-of-life metrics. Adherence will be assessed using the Simplified Medication Adherence Questionnaire (SMAQ) at three and six months, thereafter every six months.
Enrollment began October 2022 with an expected completion date of April 2025 (NCT05467176).
Presented by: Daniel Eidelberg Spratt, MD, Chairman and Professor of Radiation Oncology, University Hospitals Cleveland Medical Center, Case Western Reserve University, Cleveland, OH
Written by: Rashid Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 2023 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, San Francisco, CA, February 16th – February 18th, 2023
References:

