(UroToday.com) The 2022 GU ASCO Annual meeting included a session on novel treatment implementation focusing on PSMA targeting and beyond, with a presentation by Dr. Tanya Dorff discussing new targets in prostate cancer beyond prostate-specific membrane antigen (PSMA). Dr. Dorff notes that when defining expression potential targets, it is important to know how we measure expression and assess how much expression is enough. For immunohistochemistry, bone staining is imperfect, however Western blot, qPCR identified 100% HER2 expression in ovarian cancer (versus 29% for immunohistochemistry). Other options for defining expression of potential targets include circulating tumor cells or mRNA expression. Dr. Dorff highlighted that there are several approaches that may be used for antigen targeted therapy, including (i) antibody-drug conjugates (chemotherapy payload), (ii) antibody-drug conjugates (radioactive payload), (iii) chimeric antigen receptor T cells, and (iv) bispecific T cell engaging antibodies.
The first potential new target is prostate stem cell antigen (PSCA), which is present in 80% of prostate cancer primary tumors and 90% of metastatic lesions. PSCA is downregulated by androgen receptor suppression but overexpressed in castration resistant disease. Of note, normal tissue expression of PSCA includes in the bladder and gastrointestinal tract. PSCA targeted CAR T in mCRPC has been tested in the phase 1 setting, including radiographic and PSA responses having occurred (data being presented at GU ASCO 2022), with on-target, off-tumor toxicity manifesting as cystitis. As such, PSCA PET imaging is feasible. A second potential new target is human kallikrein 2 (KLK2), which is a serine protease with 78% homology with PSA; KLK2 is expressed in normal and malignant prostate tissue:

Dr. Dorff noted that JNJ-75229414 is a KLK2 CAR T that is being tested in an ongoing phase I trial (NCT05022849). Radiopharmaceuticals targeting KLK2 have also been reported. This includes 177Lu-hu11B6 increasing survival in prostate xenografts and Ac225-hu11B6-IgG1 successfully treating LNCaP xenografts. Dr. Dorff notes that the JNJ-69086420 (NCT04644770) dose escalation trial targeting KLK2 is ongoing.
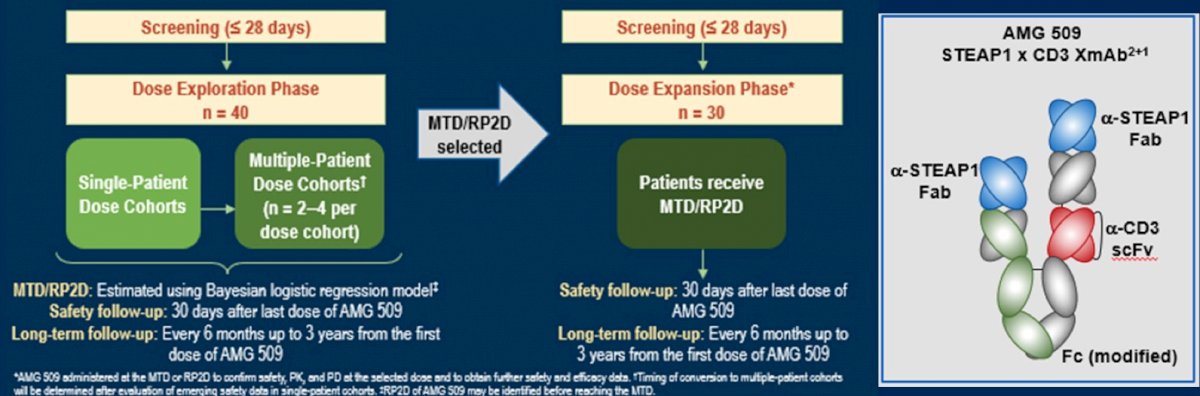
A third potential new target is STEAP-1. DSTP3086S, an antibody-drug conjugate with MMAE payload, reported 18% of patients with PSA50 response when the dose was >2 mg/kg IV q 3weeks, 6% had RECIST partial response, and 59% had circulating tumor cell conversion. Dose limiting toxicities included grade 3 transaminitis, grade 3 hyperglycemia, and grade 4 hypophosphatemia. Furthermore, a BiTE (XmAb) antibody targeting STEAP-1 (AMG509) is being tested in a phase 1 trial with the following design:
For the remainder of her presentation, Dr. Dorff discussed neuroendocrine prostate cancer, which she notes that in treatment-emergent neuroendocrine disease there are changes in antigen profiles. One target in neuroendocrine prostate cancer is DLL3, which is regulated by ASCL1, is more commonly expressed in RB1 loss, with expression acquired during treatment resistance. Furthermore, DLL3 can be heterogeneously expressed between metastases. An antibody drug conjugate targeting DLL3 has been successful in vivo and in a phase 1 trial (NCT02709889); patients with mCRPC developed lymph node metastases with undetectable PSA on treatment, with subsequent biopsy revealing small cell carcinoma. These patients received SC16LD6.5 (rovalpituzumab tesirine) therapy 0.3 mg/kg q6 weeks with responses noted on CT imaging.
A second target in neuroendocrine prostate cancer is CEACAM5. In preliminary work, 11 of 16 neuroendocrine prostate cancer samples had a high level of CEACAM5 expression, while none of the prostate adenocarcinoma lines had high-expression. Trials using CEACAM5 targeted CAR T cells have an anti-tumor effect in neuroendocrine prostate cancer cell lines, but clinically severe toxicities, including transient colitis, hepatitis, and pneumonitis.
Dr. Dorff concluded her presentation of new targets in prostate cancer beyond PSMA with the following take-home messages:
- Additional antigens show promise
- We need to understand the relationship between expression and response, and how best to measure these metrics
- We must understand efficacy/toxicity differences with different approaches (BiTE vs CAR T vs antibody drug conjugate/radiopharmaceutical) to the same antigen
Presented by: Tanya B. Dorff, MD, City of Hope Comprehensive Cancer Center, Duarte, CA
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022

