(UroToday.com) The 2022 GU ASCO Annual meeting included a renal cell carcinoma (RCC) session highlighting work from Dr. Neil Shah and colleagues presenting results of a real-world assessment of changing treatment patterns and sequence for patients with metastatic RCC in the first-line setting. Several immune-oncology (IO) agents and/or tyrosine kinase inhibitors (TKIs) have received approval for treatment of metastatic RCC in the first-line setting by Food and Drug Administration (FDA) over last few years. Limited data exists on evolving real-world treatment patterns and sequences in metastatic RCC patients receiving these agents, especially in the community oncology setting.
This study used data from The US Oncology Network of over 1,300 providers from over 480 sites across the United States from January 1, 2018 to December 31, 2020 (study period). The eligible study population included metastatic RCC patients who received:
- Ipilimumab + nivolumab (IO+IO)
- Pembrolizumab + axitinib (IO+TKI)
- Axitinib or cabozantinib or pazopanib or sunitinib (TKIs monotherapy)
in first-line setting until September 30, 2020. Descriptive statistics were used for cohort characterization.
This study identified 3,756 metastatic RCC patients, of which 1,538 were eligible including 42% (n=641) IO+IO, 18% (n=279) IO+TKI, and 40% (n=618) TKI monotherapy. The median age for the entire cohort was 67.1 years (range 25.0, 93.3), 70% (n=1,076) were male, 70% (n=1,081) were white, 38% (n=587) had BMI ≥ 30 and 79% (n=1,208) had clear cell histology. Among the overall cohort, 87% (n=1,338) had intermediate/poor risk score as per International metastatic RCC Database Consortium risk model. A summary of the baseline characteristics is as follows:
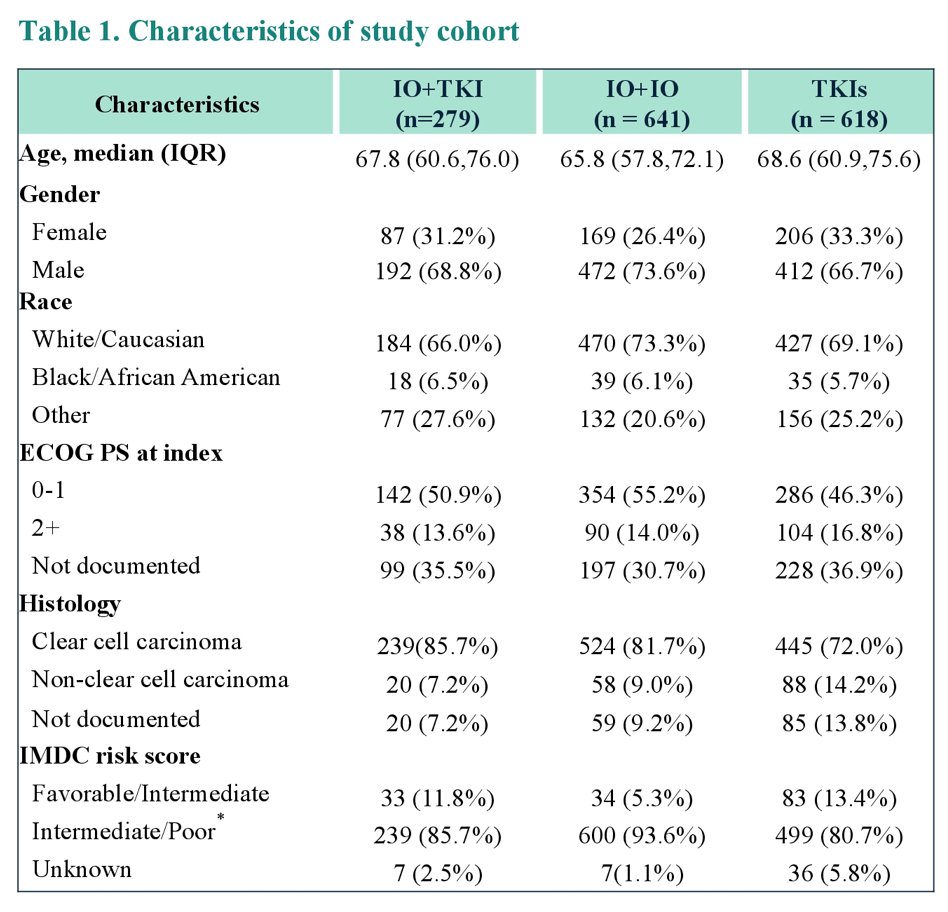
There was a notable trend towards increased utilization of IO+IO and IO+TKI following their respective FDA approvals (IO+IO: April 2018, IO+TKI: April 2019):

During the study period, metastatic RCC patients received 35% (n=535) second-line, 12% (n=184) third-line, and 4% (n=62) fourth-line treatments. Cabozantinib (49%) and pazopanib (12%), cabozantinib (51%) and ipilimumab + nivolumab (23%), and nivolumab (45%) and ipilimumab + nivolumab (20%) were the most common second-line treatments in IO+IO, IO+TKI, and TKI monotherapy cohorts, respectively:

Dr. Shah concluded his presentation of real-world changing treatment patterns for patients with metastatic RCC in the first-line setting with the following take-home messages:
- This large real-world study examined use of new FDA approved metastatic RCC treatments and their impact on treatment paradigm. Since approval of newer IO based therapy, there has been a substantial increase in the uptake of IO based combination treatments in the first-line community setting in the US
- Second-line therapies were guided by therapies in first-line with most patients receiving a TKI if they received IO-based therapy in first-line and IO-based therapy if they received a TKI in the first-line
- A longer follow-up is needed to assess their clinical impact and optimal treatment strategy in subsequent setting
Presented By: Neil Shah, MBBS, Memorial Sloan Kettering Cancer Center, New York, NY
Co-Authors: Sneha Sura, Reshma Shinde, Junxin Shi, Rodolfo F. Perini, Singhal Puneet, Nicholas J. Robert, Nicholas J. Vogelzang, Robert J. Motzer
Affiliations: Memorial Sloan Kettering Cancer Center, Ontada, Irving, TX, Merck & Co., Inc., Kenilworth, NJ, Memorial Sloan Kettering Cancer Center, New York, NY
Written By: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium, Thursday Feb 17 – Saturday Feb 19, 2022

