Dr. Gleave began by highlighting the role of molecular sub-classification of metastatic castration-resistant prostate cancer (mCRPC) to stratify treatment. In particular, he emphasized the utility of biomarkers to guide treatment choice in metastatic castration sensitive prostate cancer (docetaxel vs androgen axis-targeting agents), the optimal sequencing of androgen axis-targeting agents in castration-resistant disease, and the selection of patients for targeted or immunotherapeutic approaches.
Among these pathways, Dr. Gleave highlighted the PI3 Kinase pathway in carcinogenesis. This pathway contributes to the pathogenesis of many cancers. PTEN is a lipid phosphatase that functions to convert PIP3 to PIP2 as negative regulation of PI3K signaling. Loss of PTEN is linked with prostate carcinogenesis, progression, and treatment resistance.

Genomic alterations in the PI3K pathway are common in cancer, including, in particular, loss of the tumor suppressor PTEN gene or PI3K or AKT activating mutations. There are many mechanisms leading to PTEN inactivation such as mutation, small indels, copy number alterations, promoter hypermethylation, and RNA interference. In advanced prostate cancer (mCRPC), approximately 40% of patients have homologous deletions in PTEN.
Loss of PTEN function drives increased PI3K activity in men with mCRPC, supporting resistance to androgen receptor inhibition. Interestingly, single-agent targeting of the PI3K pathway in unselected populations of patients with CRPC and breast cancer has shown minimal activity in phase II trials.
For example, Dr. Gleave highlighted reciprocal cross-talk activation between the androgen receptor and AKT pathways may mediate prostate cancer cell survival and confer resistance to monotherapeutic interventions.

However, in murine and human xenograft models, co-targeting of these pathways is associated with response.
Among biomarker selected patients, targeting of the PI3K pathway led to improved clinical outcomes. For example, in metastatic breast cancer, pan-AKT inhibitors are associated with improved PFS and overall survival. In men with mCRPC, pan-AKT inhibitors have demonstrated benefit in phase II biomarker selected patients in combination with abiraterone or docetaxel. Dr. Gleave then highlighted data from the A.MARTIN trial which demonstrated improved rPFS in patients receiving combined ipatasertib and abiraterone among patients with PTEN loss. However, there was no benefit to the combined approach in PTEN negative patients. In the ProCAID trial, a randomized phase II study, the combination of capivasertib with docetaxel was not associated with improvements in rPFS, irrespective of biomarker status, though there were improvements in overall survival, again agnostic of biomarker status.
Dr. Gleave then discussed the phase III IPATential150 trial of receive ipatasertib (400mg PO daily) in combination with abiraterone acetate (1000mg PO daily) and prednisone (5mg PO BID) or placebo plus abiraterone acetate and prednisone in first-line mCRPC with stratification according to PTEN loss.
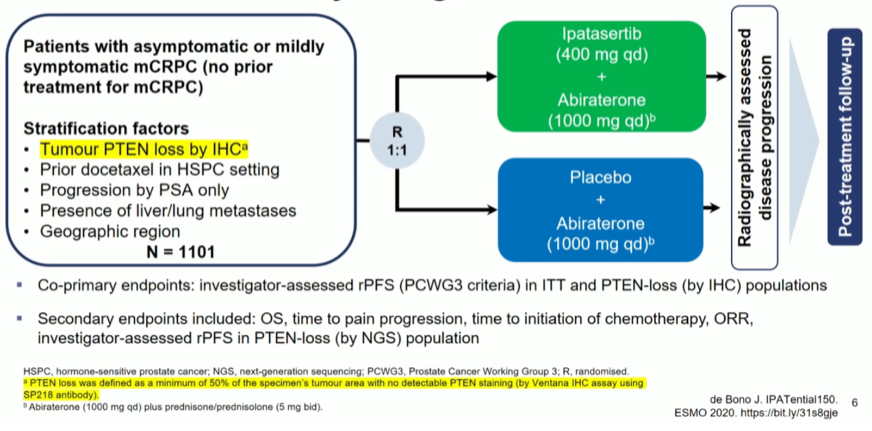
The primary endpoint was met in the PTEN population.

In both the PTEN-loss subset and the ITT population, there is a suggestion of OS benefit though the data remain immature. Notably, grade 3 and 4 events were much more common in the combined therapy arm.
Dr. Gleave then emphasized that when PTEN-loss was defined by NGS, rather than IHC, rPFS improvements were event greater.
Thus, this comes to the issue of how to characterize PI3K activation, as both immunohistochemistry (IHC) and next-generation sequencing (NGS) have their limitations.

Taken together, there are limitations to both approaches but they are potentially complementary and may be used in conjunction in composite assays. A third approach may be ctDNA assays.
In a population-based cohort of patients with mCRPC, Dr. Wyatt examined cfDNA finding that 45 of 599 patients (7.5%) had somatic activating mutations in AKT1 or PIK3CA, most of which were clonal. Notably, these mutations were rarely associated with AR copy number gain and these mutations were mutually exclusive.
In in vivo models, ipatasertib was more efficacy in patients with AKT1 mutations.
Dr. Gleave concluded that PI3K pathway activation is common in advanced prostate cancer and can drive progression and resistance. Ongoing trials are needed to refine the optimal biomarker profile and disease setting for combination therapy with AR and PI3K pathway inhibition.
Presented by: Martin Gleave, MD, FRCSC, FACS, University of British Columbia
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter during the 2021 American Society of Clinical Oncology Genitourinary Cancers Symposium (#GU21), February 11th-February 13th, 2021