San Francisco, CA (UroToday.com) There are limited effective treatments for patients with metastatic bladder cancer who are platinum ineligible. Single-agent immunotherapy has limited efficacy in an unselected population, with objective response rates (ORRs) in the 10-20% range. Thus, there is an unmet need for our patients with metastatic bladder cancer.
Enfortumab vedotin is an antibody-drug conjugate that targets Nectin-4, a protein that is highly expressed in urothelial cancer. In EV-201, patients with metastatic UC who had progressed on immunotherapy were given Enfortumab on days 1, 8, and 15 of every 28-day cycle, and the confirmed objective response rate was 44% with 12% complete responses1. Based on these results, Enfortumab has gained FDA approval in the 3rd line, for patients who have previously received an immune checkpoint inhibitor and platinum-containing chemotherapy. Recently at ESMO 2019, results from EV-103 were presented. EV-103 evaluated Enfortumab as a front-line agent in combination with pembrolizumab, and the results from cohort A (cisplatin-ineligible) demonstrated an impressive 71% objective response rate with 13% of patients achieving a complete response. This presentation provides an update on EV-103, including data on the cohort of patients who were cis ineligible.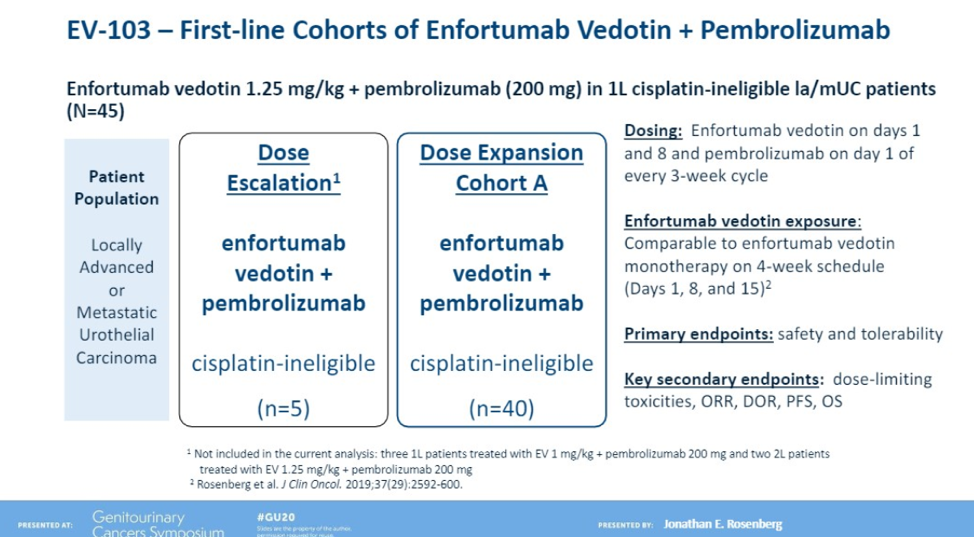
Patients were treated with EV 1.25 mg/kg on day 1 and day 9 and pembrolizumab was given day 1. Every cycle was 21 days. The primary endpoint was safety and the secondary endpoints included objective response rate and overall survival.
Data is presented on 45 patients, who have received a median of 9 cycles of therapy (range 1-22). Baseline characteristics are shown below.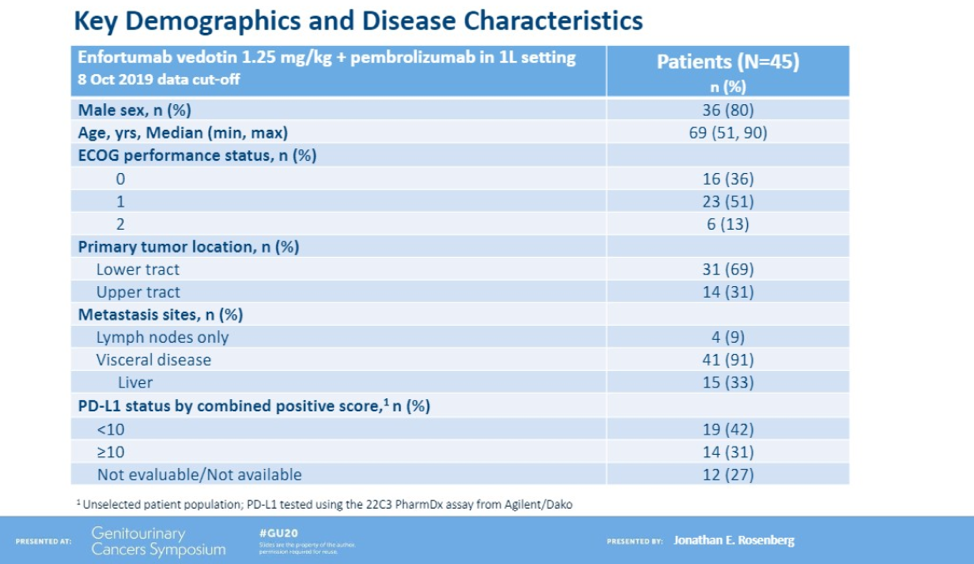
The median follow-up time is currently 11.5 months. The confirmed investigator-assessed objective response rate is currently 73.3% which is stable from the presentation at ESMO 2019, with 15.6% complete responses.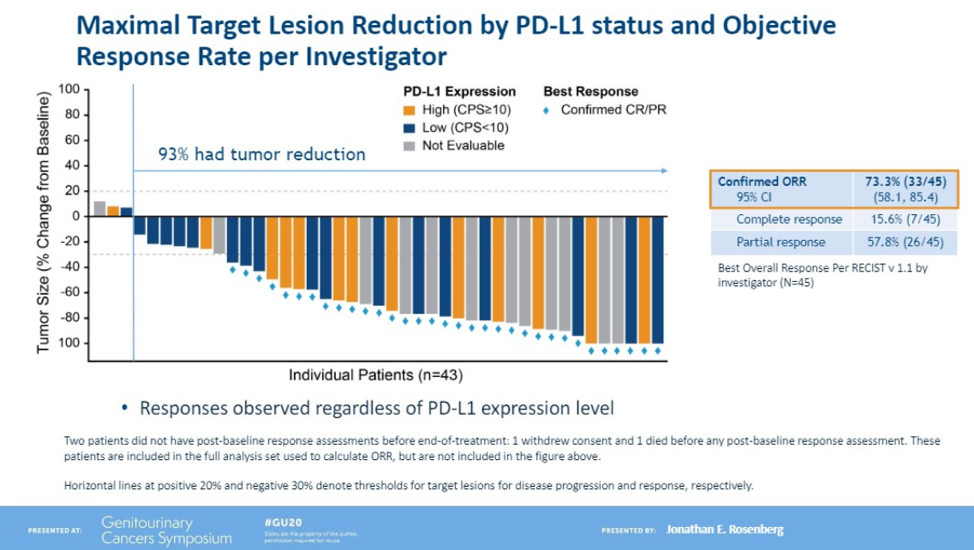
For high-risk patients with liver metastases, the objective response was 53%. There were responders regardless of PD-L1 status. Of the patients who had PD-L1 testing done, 14 patients were PD-L1 high and 19 patients were PD-L1 low, with an ORR of 78.6% and 63.2% respectively. Of the 33 responders, 55% have ongoing responses including 11 responses beyond 10 months and the median duration of response has not yet been reached.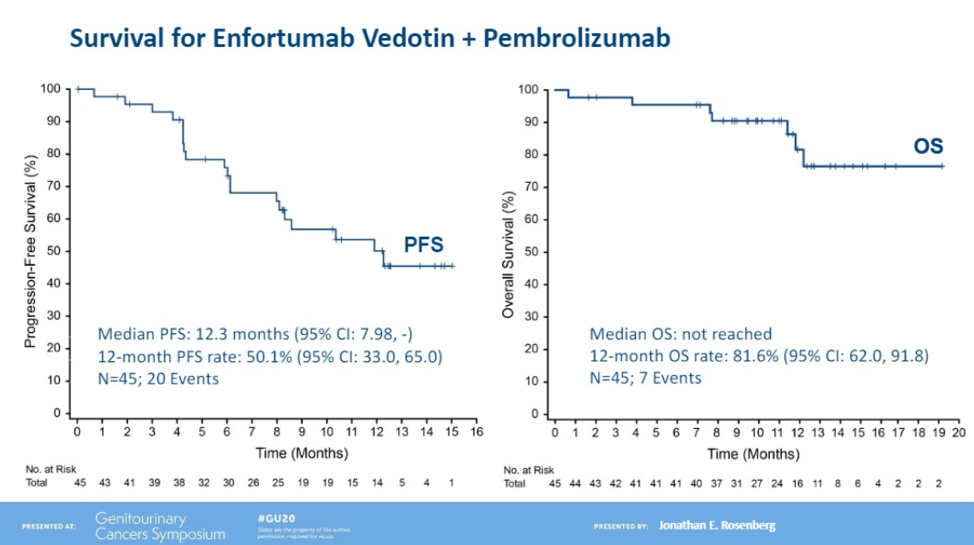

The median progression-free survival (PFS) in this cohort was 12.3 months.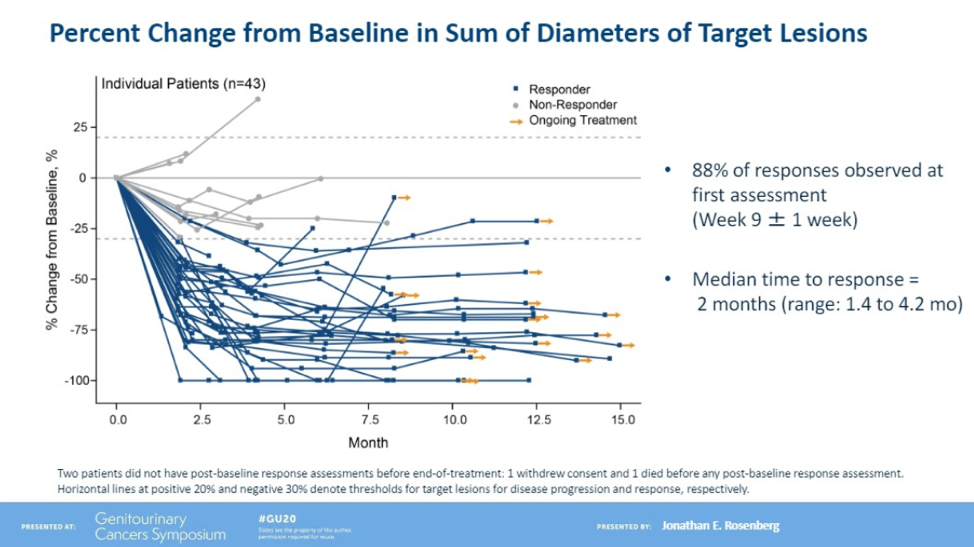
In terms of toxicity, one patient died as a result of multi-organ failure. The most common adverse events were fatigue (58%), alopecia (53%), and neuropathy (53%). 
Preliminary data is very encouraging for Enfortumab plus pembrolizumab for patients with metastatic bladder cancer, especially for platinum ineligible patients for whom there are limited therapies. Cisplatin-based regimens such as MVAC or Gemcitabine/Cisplatin historically have objective response rates between 40-50% and median progression-free survival in the 10-month range, so this regimen appears to have similar efficacy. It will be important to evaluate if a subset of these patients has long term durable responses, and additional ongoing studies are evaluating the efficacy of Enfortumab in the neoadjuvant setting in combination with chemotherapy and immunotherapy.
Presented by: Jonathan E. Rosenberg, MD, Medical Oncologist, Chief of the Genitourinary Medical Oncology Service, Division of Solid Tumor Oncology, Memorial Sloan Kettering Cancer Center, New York, NY
Written by: Jason Zhu, MD. Medical Oncologist, Division of Genitourinary Cancers, Levine Cancer Institute, Twitter: @TheRealJasonZhu, at the 2020 Genitourinary Cancers Symposium, ASCO GU #GU20, February 13-15, 2020, San Francisco, California.
References:

